We've already known that Fe+ + e Fe+, E-0.77v (1) Fe(OH)2 Fe+ + 2OH,...
Fantastic news! We've Found the answer you've been seeking!
Question:
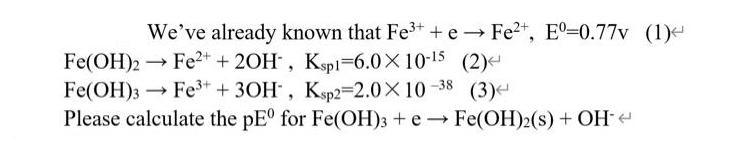
Transcribed Image Text:
We've already known that Fe³+ + e Fe²+, Eº-0.77v (1) → Fe(OH)2 → Fe²+ + 2OH, Ksp1-6.0×10-¹5 (2) -> Fe(OH)3 Fe³+ + 3OH, Ksp2=2.0X10-38 (3) Please calculate the pEº for Fe(OH)3 + e Fe(OH)2(s) + OH- → We've already known that Fe³+ + e Fe²+, Eº-0.77v (1) → Fe(OH)2 → Fe²+ + 2OH, Ksp1-6.0×10-¹5 (2) -> Fe(OH)3 Fe³+ + 3OH, Ksp2=2.0X10-38 (3) Please calculate the pEº for Fe(OH)3 + e Fe(OH)2(s) + OH- →
Expert Answer:
Answer rating: 100% (QA)
Solution To calculate the pE for the given redox reaction we need to write the balanced halfreactions and their respective standard reduction potentials E Then we can calculate the overall standard cell potential Ecell and use the Nernst equation to find the cell potential Ecell at the given conditions Finally we can use the relation pE logEcell to find the pE The oxidation halfreaction ... View the full answer

Related Book For 

Corporate Financial Accounting
ISBN: 9781337398169
15th Edition
Authors: Carl S. Warren, James M. Reeve, Jonathan Duchac
Posted Date:
Students also viewed these finance questions
-
3 / 8t = 5 / 6 t 1 / 4
-
That 1 - 1 / 2 + 1 / 3 - 1 / 4 + ... - 1 / 2( = 1 + 1 / 2 + 1 / 3 + ... + 1 / 2( - (1 + 1 / 2 + 1 / 3 + ... + 1 / n) = 1 / n + 1 + 1 / n + 2 + ... + 1 / 2n Recognize the latter expression as a...
-
Calculate Ve and Vp for the following notes. Base is 36 ft. 12 +90 C6.4 C3.6 C5.7 40.8 43.6 0 12 +30 C3.1 C4.9 C4.3 30.4 0 35.2
-
Mike and Linda are a married couple who file jointly and have $100,000 of taxable income. They have three dependent children who are full-time students in 2022. Mike and Linda provided $8,000 of...
-
The Minnesota adoption statute requires that any agency placing a child for adoption make a thorough investigation and not give a child to an applicant unless the placement is in the best interests...
-
U . S . tax liability ( before reductions for the foreign tax credit ) is $ 1 2 0 , 7 5 0 . Requirement What is Halo's foreign tax credit? What is Halo's foreign tax credit carryback or carryover?...
-
Refer to the example on page 84 but suppose the manufacturer has difficulty getting enough LED screens. Because of the shortage, the manufacturer had to obtain \(40 \%\) of the screens from the...
-
Taussig Technologies Corporation (TTC) has been growing at a rate of 20% per year in recent years. This same supernormal growth rate is expected to last for an other 2 years (g1 = g2 20%) (a) If D0 =...
-
This course is UNDC201, it includes concepts, definitions, legal provisions, different types and methods as well as markets for organized criminal activities of organized criminal groups....
-
Jaguar Plastics Company has been operating for three years. At December 31 of last year, the accounting records reflected the following: Cash Investments (short- term) Accounts receivable Inventory...
-
A 61-year-old female presents to an acute care clinic with complaints of a headache and double vision. Her erythrocyte sedimentation rate is 73 mm/hr. a) Leukocytoclastic vasculitis b)...
-
Fargo Inc. is considering a project that will require an initial investment of $1.5 million. The project will provide incremental cash inflows of $600,000 for the next five years. If the required...
-
A substantial portion of inventory owned by Prentiss Sporting Goods was recently destroyed when the roof collapsed during a rainstorm. Prentiss also lost some of its accounting records. Prentiss must...
-
Heisig Partners is considering a new project. The initial cash outflows are $1,000. The Company believes the project will yield cash inflows of $177 in year 1, $295 in year 2, $236 in year 3, and...
-
How do operating systems ensure fault tolerance and recovery in case of system failures? Discuss the role of checkpoints, journaling, and redundancy in this context.
-
1. Can you spot an example of positive self-talk and negative self-talk by Antwone Fisher? 2. How does Antwone Fisher use (or adapt) the S-TLC model to deal with his conflicts with his therapist,...
-
If the annual interest rate is 6%, would you rather have: $500 now $700 at the end of five years If you invest $1000 in a bank paying an annual interest rate of 10% compounded annually, how long it...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
In teams, select a public company that interests you. Obtain the companys most recent annual report on Form 10-K. The Form 10-K is a companys annually required filing with the Securities and Exchange...
-
Using the following data, how should the inventory be valued under lower of cost or market? Original cost ................................. $1,350 Estimated selling price ..........................
-
Reece Financial Services Co., which specializes in appliance repair services, is owned and operated by Joni Reece. Reece Financial Services accounting clerk prepared the following unadjusted trial...
-
Amherst Metal Works produces two types of metal lamps. Amherst manufactures 20,000 basic lamps and 5,000 designer lamps. Its simple costing system uses a single indirect-cost pool and allocates costs...
-
Amherst Metal Works produces two types of metal lamps. Amherst manufactures 20,000 basic lamps and 5,000 designer lamps. Its activity-based costing system uses two indirect-cost pools. One cost pool...
-
How do managers refine a costing system?

Study smarter with the SolutionInn App


