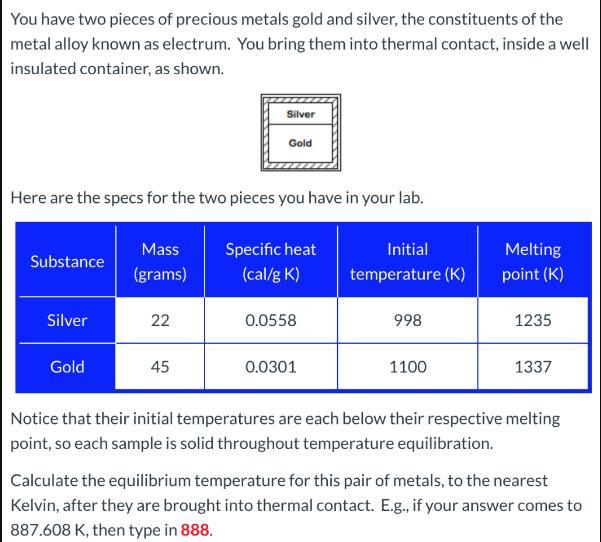
You have two pieces of precious metals gold and silver, the constituents of the metal alloy...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have two pieces of precious metals gold and silver, the constituents of the metal alloy known as electrum. You bring them into thermal contact, inside a well insulated container, as shown. Here are the specs for the two pieces you have in your lab. Substance Silver Gold Mass (grams) 22 Silver Gold 45 Specific heat (cal/g K) 0.0558 0.0301 Initial temperature (K) 998 1100 Melting point (K) 1235 1337 Notice that their initial temperatures are each below their respective melting point, so each sample is solid throughout temperature equilibration. Calculate the equilibrium temperature for this pair of metals, to the nearest Kelvin, after they are brought into thermal contact. E.g., if your answer comes to 887.608 K, then type in 888. You have two pieces of precious metals gold and silver, the constituents of the metal alloy known as electrum. You bring them into thermal contact, inside a well insulated container, as shown. Here are the specs for the two pieces you have in your lab. Substance Silver Gold Mass (grams) 22 Silver Gold 45 Specific heat (cal/g K) 0.0558 0.0301 Initial temperature (K) 998 1100 Melting point (K) 1235 1337 Notice that their initial temperatures are each below their respective melting point, so each sample is solid throughout temperature equilibration. Calculate the equilibrium temperature for this pair of metals, to the nearest Kelvin, after they are brought into thermal contact. E.g., if your answer comes to 887.608 K, then type in 888.
Expert Answer:
Answer rating: 100% (QA)
The equilibrium temperature can be found using the following equation Tequilibr... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The below figure is Figure 3 from Burgstahler and Dichev [Burgstahler, D. and Dichev, I. (1997). Earnings management to avoid earnings decreases and losses, Journal of Accounting and Economics 24(1):...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
According to the 1980 revisions to the Copyright Act of 1976,_____. a. the owner of a copyrighted program is allowed to make multiplecopies or adaptations for use on other computers b. copyright...
-
A solid copper disk of radius R rotates at angular velocity w in a perpendicular magnetic field B. The figure shows the disk rotating clockwise and the magnetic field into the page. (a) Is the charge...
-
your student club is organising a community service for Diamond Heart Orphanage and you have been elected as the person in charge for finance. Write a letter to Baba Caterer to request for food sponso
-
How often do you think boards should evaluate their own performance? Why?
-
Lynch Co. uses the percentage of receivables basis to record bad debts expense and concludes that 2% of accounts receivable will become uncollectible. Accounts receivable are $400,000 at the end of...
-
Why does external analysis usually start with the macro - environment & proceed to industry analysis? A ) This is just a convention that has developed over time. It does not matter what order the...
-
David R. and Ella M. Cole (ages 39 and 38, respectively) are husband and wife who live at 1820 Elk Avenue, Denver, CO 80202. David is a self-employed consultant, specializing in retail management and...
-
Sunland Company is constructing a building. Construction began in 2017 and the building was completed 12/31/2017. Sunland made payments to the construction company of $3102000 on 7/1, $6432000 on...
-
1. Bank balance as shown in the bank statement, P 648,364. Cash in Bank balance in the books of the business, P 707,404. Deposit in transit is P46,800. Checks outstanding for the month: No. 15600...
-
John decide to save to buy a car in three years time. He will invest a fixed sum into a three - year fixed deposit savings account that pays interest at 9 % p . a . how much would he need to invest...
-
Java Programming how to write test strategy for the Fox class inheriate from parten class animal. public class fox extends Animal { public Bilby(String id,boolean isNewBorn) { super(id, true,...
-
Suppose you plan to invest $ 1 , 2 0 0 today at an interest rate of 1 0 % . How long will it take to get $ 1 , 5 0 0 from the investment? How long would it take $ 1 , 0 0 0 to double if it were...
-
How can the technology used in IBM Watson give a competitive advantage to an organization? Think of your current workplace or an organization you worked for in the past. How can the technology used...
-
A roller-coaster car has a mass of 508 kg when fully loaded with passengers. The path of the coaster from its initial point shown in the figure to point B involves only up-and-down motion (as seen by...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Use bond energies to estimate H for the reaction H2(g) + O2(g) H2O2(g)
-
In the following four models, C atoms are black, H atoms are light blue, O atoms are red, and N atoms are dark blue: a. Write the molecular formula of each molecule. b. Write the condensed structural...
-
Sodium-23 is the only stable isotope of sodium. Predict how sodium-20 will decay and how sodium-26 will decay.
-
An increase in the demand for land will _________ the quantity supplied and _________ the price of land.
-
The supply of land is perfectly _________.
-
_________ is the price paid for land or any other factor that has a fixed supply.

Study smarter with the SolutionInn App


