Show the consistency of the exchange energy formula (8.12.15) and the result of integrating (8.12.3). Fock(k) =
Question:
Show the consistency of the exchange energy formula (8.12.15) and the result of integrating (8.12.3).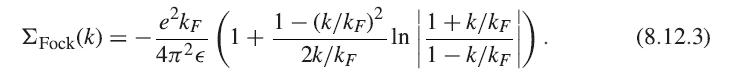

Transcribed Image Text:
ΣFock(k) = - e²kF 4л²€ 1+ 1 - (k/kF)² 2k/kF -In 1+k/kF 1 - k/kF (). (8.12.3)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
In order to prove the statement we must calculate the exchange ene...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
Accounts Receivable AED 5,000 Accounts Payable 15,000 Advertising Expense 2,000 16,500 Building Cash Common Stock 50,000 Dividends 1,200 Equipment 2,000 Land 70,000 Notes Payable 60,000 3,400 Office...
-
Tatum Company has four products in its inventory. Information about the December 31, 2011, inventory is as follows: The normal gross profit percentage is 25% of cost. Required: 1. Determine the...
-
Discuss why the user interface is important in an accounting system.
-
Identify how ethical investment can affect corporate decision making regarding sustainable business operations.
-
Multiple-Choice Questions 1. A business owner makes 1,000 items a day. Each day he or she contributes eight hours to produce those items. If hired, elsewhere he or she could have earned $250 an hour....
-
Trying to break an encryption key by trying every possible combination of characters is called what? 1 point A social engineering attack A brute force attack A rainbow table attack A known cyphertext...
-
According to (4.8.15) in Section 4.8, the scattering rate for fermions is proportional to 1/2 (U D U E ) 2 , where UD is the direct interaction vertex and U E is the exchange vertex. In the diagram...
-
Show that the Matsubara Greens function is periodic, that is, for prove Use the cyclic property of traces (8.13.10), above. G(T T) = -i(T(az(t)a/(t)))T, k
-
The Doppler broadening in a gas can be expressed as where M is the molar mass. For the sodium transition, ν 0 5.0933 à 10 14 s -1 . Calculate Îν and...
-
How was the interest calculated ? 5. The Fredon corporation has recently sold Ron Obvious a note which pays $5000 a year from today. If Ron paid $4800 for this note, the effective interest rate on...
-
Provide your insights about the importance of cultural competence and diversity consciousness in the allied health care profession. How will you incorporate these skills into your future career?
-
Two different textbook definition of the meaning of London as a global city and headquarters of major global corporations and investments. Quantitative source data of comparable nature on London's...
-
The following information relates to Blake Auto Supply. Net Sales Revenue Cost of Goods Sold Interest Revenue Operating Expenses $194,000 150,000 12,000 42,500 Calculate the net income of Blake Auto...
-
Using a lever, Daisy is able to lift a 1 0 , 0 0 0 N car off the ground with a force of 1 0 0 0 N . What is the mechanical advantage of the lever?
-
In which polymer, polyethylene or polyvinyl chloride, would you expect to find rhe stronger intermolecular forces, assuming that the average chain lengths are equal?
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the...
-
The Waffle House has $25 million of risk-free debt outstanding. This debt has an annual coupon rate of 8% and matures in 2 years. Coupons are paid once per year. The current term structure is flat at...
-
Calculate the perihelion and aphelion velocities (km/s) of an object with an aphelion of 3.0 AU and perihelion of 1.0 AU.
-
1 . A company purchases new cement manufacturing assets that cost $ 1 8 million. This is classified in the 1 5 - year property class using MACRS - GDS . What would be the depreciation allowance at...

Study smarter with the SolutionInn App


