Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-:
Question:
Calculate the formal charges on the atoms shown inred.
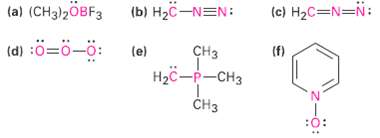
Transcribed Image Text:
(a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :ö=ö-ö: (e) сHз (f) Нас —р-снз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
To save space molecules are shown as linebond structures with lone pairs rather than as electrondo...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species: a) H H-C-0_H d) H H C=N=N: b) H H-C-0: f) SF: : | H-NC-C- ...
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
There are 38 numbers in the game of roulette. They are 00, 0, 1, 2, . . ., 36. Each number has an equal chance of being selected. In the game, the winning number is found by a spin of the wheel. Say...
-
Ricky owns stock in Dove Corporation. His adjusted basis for the stock is $90,000. During the year, he receives a distribution from the corporation of $75,000 that is labeled a return of capital...
-
A 100.0 mL solution of 0.0500 M Mn buffered to pH 9.00 was titrated with 0.0500 M EDTA. (a) What is the equivalence volume, Ve, in milliliters? (b) Calculate the concentration of Mn+ at V = 1/2 Ve+....
-
The online review service Yelp helps millions of consumers find the goods and services they seek. To help consumers make more-informed choices, Yelp includes over 120 million reviews. The file yelp...
-
1. Are there any disagreements between or among the resulting orderings? If so, why do you think that is the case? 2. What can be done to mitigate any observed disagreement between or among the...
-
In the realm of clinical mental health counseling, the roles of consultation, advocacy, and evaluation play pivotal roles in ensuring comprehensive and effective client care. How do you perceive the...
-
Poller Corporation (a fictional company) operates a chain of discount retail stores. The follo ing is information taken from a recent Potter annual report. Note 5: Mortgages on Property, Plant, and...
-
Methanethiol, CH2SH, has a substantial dipole moment ( = 1.52) even though carbon and sulfur have identical electro negativities. Explain.
-
Which of the following pairs of structures represent resonanceforms? (b) :0: (a) :0: and and (d) :: (c) :0: :0: and and
-
Shelly Inc. bonds have a 6 percent coupon rate. The interest is paid semiannually, and the bonds mature in 8 years. Their par value is $ 1,000. If your required rate of return is 4 percent, what is...
-
Foreign Direct Investment and Cross-Border Acquisitions Project Description: While making a decision about investing abroad a company has to consider a wide range of factors comprising the investment...
-
What organizational capabilities are required to support the launch of "Command Products"?
-
Discuss how positive psychology can be used for the greater good. Some assert that positive psychology represents the narcissistic tendencies of our culture. Explain how positive psychology can be...
-
Explore leverage as it related to the financial crisis of 2008/2009 and share what you learned?
-
QUESTION 4 We can make a uniform Electric field by spreading opposite charges on large-Area metal plates that aren't very far apart, like an Oreo. Suppose the two plates are 0.200[m]x0.200[m], set...
-
Recommend a solution that will provide the fund manager the opportunity to earn currency alpha through active foreign exchange management. Gupta and the fund manager of Portfolio A discuss the...
-
Question 6.10 Current and deferred tax worksheets and tax entries From the hip Ltd?s statement of profit or loss for the year ended 30 June 2007 and extracts from its statements of financial position...
-
The following equation of state has been proposed for a fluid where B and C are constants. a. Does this fluid exhibit a critcal point? Prove it. b. If you believe the answer to part a. is yes, derive...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
Privack Corporation has a standard cost system in which it applies overhead to products based on the standard direct labor-hours allowed for the actual output of the period. Data concerning the most...
-
HomeLife Life Insurance Company has two service departments ( actuarial and premium rating ) and two production departments ( advertising and sales ) . The distribution of each service department s...
-
Otsego Industries manufactured 300,000 units of product last year and identified the following costs associated with the manufacturing activity: Variable costs: Direct materials used Direct labor...

Study smarter with the SolutionInn App


