The reaction of ethyl butanoate with sodium ethoxide in CH 3 CH 2 OH gives (a) (b)
Question:
The reaction of ethyl butanoate with sodium ethoxide in CH3CH2OH gives
(a)
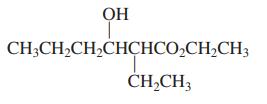
(b)
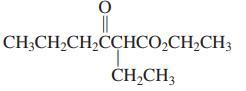
(c)
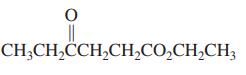
(d)
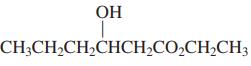
Transcribed Image Text:
ОН CH;CH,CH,CHCHCO,CH,CH3 ČH,CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Base catalysed condensation of same ester containing alfa hydrogen atom is ...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Compound A (C7H13Br) is a tertiary bromide. On treatment with sodium ethoxide in ethanol, A is converted into B (C7H12). Ozonolysis of B gives C as the only product. Deduce the structures of A and B....
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
Give the structure of the expected product from the reaction of isopropyl benzene with (a) Hydrogen (3 mol), Pt (b) Sodium and ethanol in liquid ammonia (c) Sodium dichromate, water, sulfuric acid,...
-
If you sign up for a savings account with an annual interest rate of 12% (1% per month) that you will receive 4 million won in 2 years, what is the monthly amount you have to pay? However, suppose...
-
Consider the two economies shown in Figure 3.1. When there is free trade, are we sure that each country should specialize completely in producing only one of the products? For instance, perhaps each...
-
Debate the differences between an alert-based decision-support management-accountability budgeting, monitoring, and reporting system and a standard reporting system that does not provide instant...
-
In September 2013, Ugo Mattera entered into a written construction contract with Baja Properties, LLC. Stephen Chad Golden, the sole owner of Baja Properties, signed the contract and addendums on...
-
Erie Company manufactures a small CD player called the Jogging Mate. The company uses standards to control its costs. The labor standards that have been set for one Jogging Mate CD player are as...
-
decribe the health sttsus of unit members (if any of the family describe below)Mother (29yo) and Father (32yo) 2 children (1 elementary, 1 high school) Origin: Mohawk (from Kahnawake) Address: 295,...
-
Allied Food Products is considering expanding into the fruit juice business with a new fresh lemon juice product. Assume that you were recently hired as assistant to the director of capital budgeting...
-
Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these have K a greater than methanol). Which ones? (a) A and B; (b) B and C; (c) C and D; (d) D and A; (e) D and B....
-
When acid A (margin) is heated to 230C, CO 2 and H 2 O are evolved and a new compound is formed. Which one? (a) (b) HO 2 CCH 2 CH 2 CH 2 CH 3 (c) (d) CH 3 CH 2 CH(CO 2 H) 2 (e) CO,H HO,C(CH2),CH CO,H
-
How should the joint venturers share of assets in the joint venture be disclosed?
-
Azalea Seafood Gumbo Shoppe in 2004 John E, Gamble Urlif'CiNily OrS.01,11/7 Vahaina John Addison had just returned from lunch when his partner. Mike Rathle, remarked that Wal-Mart's weekly order for...
-
What is stage four of the informed citizen to the editorial and make a final judgment on whether or not the editorial violates the standards for the informed citizen. The claim there is a strong case...
-
You have introduced a standard cost accounting system for FOL. The following standard costs have been developed for the Cattle drench. Direct materials (1 kilogram) $ 20 Direct Labour (.25 hours) $...
-
A typesetter, on the average, makes one error in every 500 words typeset. A typical page contains 300 words. What is the probability that there will be no more than two errors in five pages? I am...
-
The unique military culture, for example, unit cohesion, concern for reputation, never leave a man behind, stoicism, how could this present a barrier in entering counseling and being successful in...
-
The IRS presumes that two types of workers should be classified as independent contractors. Who are these workers, and why is the automatic classification justified?
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
Assign formal charger to the atoms in each of the followingmolecules: ( NNEN: CH (c) HCN3DN-DN: (a) H3C-N-O: CH
-
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain - C-OH - C=C c=C ...
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
2) What is the rate of return in the following cash flow? -4816 +1000 +2000 +3000
-
3. Assume that the manufacturing of generic medicine XYZ is a perfectly competitive industry. Suppose that the annual demand for this medicine is given by: Q = 1,450 - 30P. There are exactly a...
-
Suppose that a consumer's utility is given by 1 MU = y 4x - 3 1 ' - 3 3y respectively. 1 2 - - u(x,y) = 2x 3 y " 3 in which case the marginal utilities will be given by MU = 23 2y and x 2 3x 3...

Study smarter with the SolutionInn App


