An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ]
Question:
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag+] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in a solution of 5.0 M NH3 that is also 0.010 M in Cu(NH3)42+, what is the cell potential at 25οC?
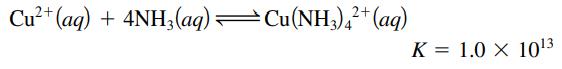
Transcribed Image Text:
Cu²+ (aq) + 4NH3(aq) —Cu(NH3)4²+ (aq) K = 1.0 X 10¹3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The cell potential at 25C Ecell Ag10 M 00060 M Cu2aq50 M0010 ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ] = 1.00 M separated by a porous disk from a compartment with a copper metal electrode immersed in a...
-
An electrochemical cell consists of a nickel metal electrode immersed in a solution with [Ni 2+ ] = 1.0 M separated by a porous disk from an aluminum metal electrode. a. What is the potential of this...
-
Gooran, Inc., has current assets of $240 million; property, plant, and equipment of $380 million; and other assets totaling $120 million. Current liabilities are $170 million and long-term...
-
How could a system be designed to allow a choice of operating systems from which to boot? What would the bootstrap program need to do?
-
Ethane gas (C2H6) at 25°C is burned in a steady-flow combustion chamber at a rate of 5 kg/h with the stoichiometric amount of air, which is preheated to 500 K before entering the combustion...
-
Determined to reduce the \(\$ 7\) per week cost associated with heat loss through their patio window by convection and radiation, the tenants of Problem 9.15 cover the inside of the window with a...
-
When a movement was organized to build Charles City College, Hauser and others signed pledges to contribute to the college. At the time of signing, Hauser inquired what would happen if he should die...
-
1. Two crates with treasure, a small one with mass m=110kg and a large one with mass M=329kg are connected by a massless rope and are on a frozen lake which you may assume to be frictionless. A rope...
-
Velocity profiles and the stress components ij. For each of the following velocity distributions, draw a meaningful sketch showing the flow pattern. Then find all the components of 'r and pvv for the...
-
Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (K sp ) for CdS given the following standard reduction potentials: CdS (s) + 2e ...
-
Consider a concentration cell that has both electrodes made of some metal M. Solution A in one compartment of the cell contains 1.0 M M 2+ . Solution B in the other cell compartment has a volume of...
-
The following information was taken from Riddell Ltds adjusted trial balance as at April 30, 2016: Sales revenue.................... $1,045,800 Interest revenue....................... 7,200 Sales...
-
Explain what the Per Process Table of Open Files of a process is in the classical Unix system. With the help of a C program fragment, show how the standard error of a process may be redirected away...
-
A partial trial balance for the Ryman Corporation is presented in the table below. Ryman has recorded some, but not all, of its transactions for the current reporting period, the year ending December...
-
Create one practical SMART goal for the company inthe major project. Who is the major competitor for your company in your major project? Using Porter's National Diamond theory , why is your...
-
Suppose that Fred wakes up earlier than Barney and decides how many pounds of cod to catch and calls the dock manager to tell him how many pounds of cod to bring to dock. When Barney wakes up, he...
-
Derive the differential equation of motion for the U-tube manometer shown below and obtain the period of oscillations. You may assume that the viscosity of the liquid is negligible. Denote the...
-
In Exercise use an adjacency list to represent the given graph. b.
-
Evaluate the function at the given value(s) of the independent variable. Simplify the results. (x) = cos 2x (a) (0) (b) (- /4) (c) (/3) (d) ()
-
A bowling ball has a weight of 12 lb and the length of the lane is approximately 60. feet. Treat the ball in the lane as a one-dimensional box. What quantum number corresponds to a velocity of 7.5...
-
For a particle in a two-dimensional box, the total energy eigenfunctions are a. Obtain an expression for E nx , n y in terms of n x , n y , a, and b by substituting this wave function into the two...
-
Consider the contour plots of Problem P15.17. a. What are the most likely area or areas Îx Îy to find the particle for each of the eigenfunctions of HË depicted in plots af? b. For...
-
What are the vulnerabilities of embedded operating systems, such as, but not limited to, IoT devices, programmable logic devices, and vehicle control systems.
-
Factor the polynomial completely. 7x-17x-12
-
What are some relevant recommendations about Coca-Cola's Planning, Design, and Implementation of an Enterprise Resource Planning System that other companies can apply to their own initiatives?

Study smarter with the SolutionInn App


