Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (K
Question:
Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (Ksp) for CdS given the following standard reduction potentials:
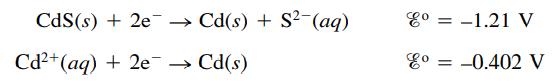
Transcribed Image Text:
CdS (s) + 2e →→ Cd(s) + S² (aq) Cd²+ (aq) + 2e → Cd(s) ४० = -1.21 V co = -0.402 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Thorough solution with explanation The solubility product ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
For the circuit in Fig. 6.70, calculate the value of R that will make the energy stored in the capacitor the same as that stored in the inductor under dc conditions. 160 uF 5A 2 4 mH
-
Calculate the value of the consumption function at each level of disposable income in Table 1 if a = 100 and mpc = 09.
-
Calculate the value of a $1,000 bond which has 10 years until maturity and pays quarterly interest at an annual coupon rate of 12 percent. The required return on similar-risk bonds is 20 percent.
-
The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: Dec. 31, 20Y9 Dec. 31, 20Y8 Assets Cash $70,720 $47,940 Accounts receivable (net) 207,230 188,190...
-
Suppose you are a depositor at Melvin's Bank, which has the balance sheet shown in Table 10.1A. Deposit insurance does not exist. You originally deposited your money in Melvin's Bank because its...
-
Consider the following cash flows of two mutually exclusive projects for AZ-Motorcars. Assume the discount rate for both projects is 10 percent. a. Based on the payback period, which project should...
-
Does the fact that selling is included as part of the promotional mix weaken or strengthen its role as a sub-element of marketing?
-
The following defined pension data of Doreen Corp. apply to the year 2012. Defined benefit obligation, 1/1/12 (before amendment) ... $560,000 Plan assets, 1/1/12 ................... 546,200 Pension...
-
The adjusted trial balance of Gibson Company for the year ended December 31, 2024, is as follows: Cash Accounts Receivable Notes Receivable Merchandise Inventory Equipment Allowance for Doubtful...
-
Jennifer Daoust is reading the documents prepared by the members of the team working on the audit of receivables for a large client. Jennifer is the senior manager assisting the engagement partner,...
-
The electrolysis of BiO + produces pure bismuth. How long would it take to produce 10.0 g Bi by the electrolysis of a BiO + solution using a current of 25.0 A?
-
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in...
-
The data file freddie 2 contains monthly housing price indices for the prices of houses in ChampaignUrbana, Illinois (CHURB), and Charlottesville, Virginia (CHARV) from 1982M1 to 2015M12. a. Plot the...
-
Maya Company currently buys a component part for $5 per unit. Maya estimates that making the part would require $2.30 per unit of direct materials and $1.10 per unit of direct labor. Maya normally...
-
How do demographic shifts and changing consumer preferences intersect to shape market trends in advanced markets ?
-
In the ICD-10-CM Alphabetic Index, which word would you look under as your main term? Hashimoto's disease
-
What role does disruptive innovation play in driving market trends within the landscape of advanced industries ?
-
How are emerging paradigms such as the sharing economy and circular economy models influencing market trends within advanced sectors, and what are the implications for traditional business models ?
-
How much heat is required to change 1.0 kg of ice, originally at 20.0C, into steam at 110.0C? Assume 1.0 atm of pressure.
-
Determine the values of the given trigonometric functions directly on a calculator. The angles are approximate. tan 0.8035
-
Reaction of iodoethane with CN yields a small amount of isonitrile, CH3CH2N C, along with the nitrile CH3CH2C N as the major product. Write electron-dot structures for both products, assign formal...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain. CH3CH2CHCH3 2-Butanol
-
(a) What is the kinetic energy in joules of a 1140-kg automobile traveling at 93 km/h? (b) How much work would have to be done to bring a 1140-kg automobile traveling at 93 km/h to a stop? 4. How...
-
A charging RC circuit controls the intermittent windshield wipers in a car. The emf is 12.0 V. The wipers are triggered when the voltage across the 120F capacitor reaches 12.0 V; then the capacitor...
-
A block of mass 2 kg is attached to a spring with a spring constant of 100 N/m. The block is released from rest and moves along a rough horizontal surface with a coefficient of friction of 0.2. How...

Study smarter with the SolutionInn App


