Consider Figure 11-5. For an equal component alloy of A and B starting at 1400C, write each
Question:
Consider Figure 11-5. For an equal component alloy of A and B starting at 1400°C, write each reaction as the crucible is cooled to room temperature (25°C).
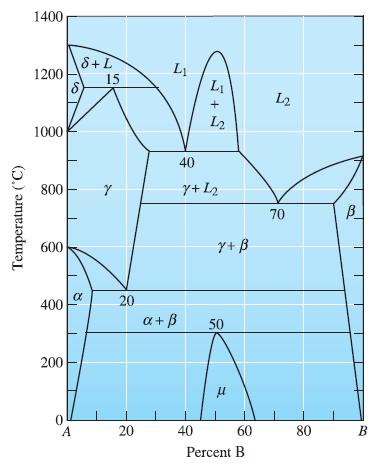
Transcribed Image Text:
Temperature (°C) 1400 1200 1000 800 600 400 200 0 S A S+L α 15 Y 20 20 Li a + ß L₁ + 12 40 7+12 40 Y+ B 50 μ 60 Percent B L2 70 80 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Reactions are as follows 1 L 1 930 0 C ...View the full answer

Answered By

User l_831638
Hello! My name is Sadhana.M.S. I made specilization in Nuclear Physics at Kuvempu university.I've been an physics tutor for degree level student . My motto is "teach student how to learn".
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
For what types of A and B is the simple assignment statement A = B legal in C++ but not Java?
-
For the separation of A and B by column 22 in the previous problem: a. If broadening is mainly due to longitudinal diffusion, how should the flow rate be changed to improve resolution? b. If...
-
Before any reaction occurs, the concentrations of A and B in the reaction below are each 0.03900 M . What is the equilibrium constant if the concentration of A at equilibrium is 0.01365 M ? 2A(aq) +...
-
Can public works increase equilibrium wages?
-
Calculate the forward discount or premium for the following spot and three-month forward rates: (a) SR = SF2/1 and SF2.02/1 where SF is the Swiss franc and is the euro (b) SR = 200/$1 and FR = 190/$1
-
Part 1. The Coca-Cola Company's (CCC) balance sheet reports the asset Goodwill. Assume that CCC purchased an asset to be included in Goodwill as part of the acquisition of another company, which...
-
Pleading rules are principally contained in the California Code of Civil Procedure. However, in addition to the CCP, local rules generally control mechanics. Find some special local rules that are...
-
The balance sheet of Aqua, Inc., a world leader in the design and sale of telescopic equipment, reported the following information on its balance sheets for 2018 and 2017 (figures are in thousands):...
-
Pik-Fast, a chain of convenience stores, is planning to open a new store. The activities required, their immediate predecessors, and the optimistic, most likely, and pessimistic estimates of their...
-
During the first quarter of 2018, Berkshire Hathaway purchased 75 million shares of Apple, Inc.'s stock. For illustration purposes, assume that all shares were purchased on January 2, at the closing...
-
Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition of the first solid to form during solidification; (c) The amounts and compositions of each...
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Use the information from Brief Exercise 9-49. On January 1, 2020, Mayor Company issued 40,000 5-year bonds with a $1,000 par value each. The market values the bonds at $30,000 less than the face...
-
See the Winter Sports Inc. data from S8-2. Assume that Winter Sports reputation has diminished and other resorts in the vicinity are charging only \($65\) per lift ticket. Winter Sports has become a...
-
Each morning, Jamal Imery stocks the drink case at Jamals Beach Hut in Charlotte, North Carolina. Jamals Beach Hut has 110 linear feet of refrigerated display space for cold drinks. Each linear foot...
-
What must be the tension applied to a \(12-\mathrm{m}\) plastic string weighing \(43.5 \mathrm{~N}\) to produce a wave that propagates at a speed of \(25 \mathrm{~m} / \mathrm{s}\) ?
-
Air at a stagnation pressure of 1 MPa flows isentropically through a CD nozzle and exhausts into ambient at 40.4 kPa. The edge of the jet, as it comes out of the nozzle is deflected by 18 (counter...
-
InteliSystems manufactures an optical switch that it uses in its final product. InteliSystems incurred the following manufacturing costs when it produced 70,000 units last year: InteliSystems does...
-
Define stock split. How does a stock split differ from a stock dividend?
-
Define cultural intelligence. Cite the books or journal articles you found in Capella's library. Explain why cultural intelligence is important for HR practitioners and other organizational managers.
-
Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O 4 2- ) without precipitating 0.010 M Ca 2+ ? CaC204 Ksp = 1.3 x 10-8 %3D Ce,(C,O4)3 Ksp 5.9 X 10-30
-
For a solution of Ni 2+ and ethylenediamine, the following equilibrium constants apply at 20C: Calculate the concentration of free Ni 2+ in a solution prepared by mixing 0.100 mol of en plus 1.00 mL...
-
If each compound is dissolved in water, will the solution be acidic, basic, or neutral? (a) Na Br (e) (CH3),N CI (b) Na CH,CO, (f) (CH,),N*O-co, (c) NH CI (g) Fe(NO3)3 (d) K3PO4
-
This is for my Organizational Behavior class. This is due by midnight tonight, July 30. Please help!! The organization I have chosen is a rural health care facility...a clinic. What are the basic...
-
What are the functions of organizational culture? Use your own experience in an organization (workplace, school, gym, etc.) to explain how organizational culture contributes either positively or...
-
In 2 0 2 4 , Bratten Fitness Company made the following cash purchases: The exclusive right to manufacture and sell the X - Core workout equipment from Symmetry Corporation for $ 2 1 8 , 0 0 0 ....

Study smarter with the SolutionInn App


