Consider only the transitions involving the first four energy levels for a hydrogen atom: a. How many
Question:
Consider only the transitions involving the first four energy levels for a hydrogen atom:
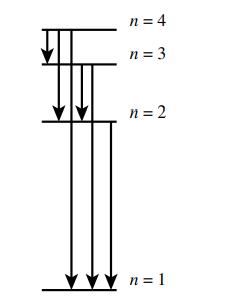
a. How many emissions are possible for an electron in the n = 4 level as it goes to the ground state?
b. Which electronic transition is the lowest energy?
c. Which electronic transition corresponds to the shortest wavelength emission?
Transcribed Image Text:
n = 4 n = 3 n = 2 n=1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Solution a The lowest energy transition is the 1S32 2S12 transition for which there are two possible ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
A hydrogen atom has an electron in the n = 5 level. (a) If the electron returns to the ground state by emitting radiation, what is the minimum number of photons that can be emitted? (b) What is the...
-
The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is the contribution to the average energy from electronic degrees of freedom for V when T =...
-
An excited hydrogen atom with an electron in the n = 5 state emits light having a frequency of 6.90 1014 s-1. Determine the principal quantum level for the final state in this electronic transition.
-
In a given week, 12 babies are born in hospital. Assume that this sample came from an underlying normal population. The length of each baby is routinely measured and is listed below (in cm): 49, 50,...
-
For a new product, sales volume in the first year is estimated to be 80,000 units and is projected to grow at a rate of 4% per year. The selling price is $ 12 and will increase by $ 0.50 each year....
-
Suppose X ~ N(4, 2). What value of x is two standard deviations to the right of the mean?
-
After closing its accounts at October 31,2010, Simmons Realty, Inc., had the following account balances: Prepare Simmons Realtys post-closing trial balance at October 31,2010. List accounts in proper...
-
Soft phonon mode consider a line of ions of equal mass but alternating in charge, with e p = e(?1) p as the charge on the pth ion the inter atomic potential is the sum of two contributions (1) a...
-
Brierly Ltd is a very profitable and wealthy listed company which operates several bars and restaurants across Victoria and New South Wales. Alex, Henry, Beatrice, Sara and Eugenie are the company's...
-
The statements of financial position of Lochnagar Ltd for the past two years and the company's most recent income statement are set out below. Income statement for the year to 31 October 2018. ? ? ?...
-
In the 1930s, electron microscopes were first used to provide structural details of human cells. If an electron microscope uses electrons having a wavelength of 0.1 nm, calculate the velocity of the...
-
A particle has a velocity that is 90.% of the speed of light. If the wavelength of the particle is 1.5 10 -15 m, calculate the mass of the particle.
-
Why are so many multinational companies using short-term international assignments (less that 1 year in length)? Which would you prefer, a long- and short-term international assignments?
-
A solution of \(13.5 \%\) by weight of polyisoprene has the following power-law parameters: \(\Lambda=5000 \mathrm{~Pa} \cdot \mathrm{s}^{n}\) and \(n=0.2\). Consider the flow of such a solution in a...
-
A mixture of titanium $(\mathrm{SG}=4.5)$ and silica $(\mathrm{SG}=2.65)$ particles, with diameters ranging from 50 to $300 \mu \mathrm{m}$, is dropped into a tank in which water is flowing upward....
-
After blowing up a balloon, you release it without tying off the opening, and it flies out of your hand. If the diameter of the balloon is 6 in., the pressure inside it is $1 \mathrm{psig}$, and the...
-
A speedboat is propelled by a water jet motor that takes water in at the bow through a $10 \mathrm{~cm}$ diameter duct and discharges it through a $50 \mathrm{~mm}$ diameter nozzle at a rate of $80...
-
A water stream contacts a bed of particles, which have diameters ranging from 1 to $1000 \mu \mathrm{m}$ and $S G=2.5$. The water stream flows upward at a rate of $3 \mathrm{~cm} / \mathrm{s}$. What...
-
During the fourth quarter of 2016, Zinner, Inc., generated excess cash, which the company invested in trading securities as follows: 2016 Nov 17 Purchased 1,300 common shares as an investment in...
-
Find the inverse, if it exists, for the matrix. -1
-
Indicate whether each of these objects is chiral or achiral: (a) Golf ball (b) Baseball glove (c) Clock (d) T-shirt (e) Dress shirt (f) Automobile
-
Determine whether each of these molecules is chiral, for those that are chiral, put an asterisk at the chirality center. b) a) d) I e) )
-
Indicate whether each of these objects or molecules has a plane of symmetry: c) Ear b) Pencil a) Idealized human face I CH3 e) f) d) CH3 . Cl Br CH3 "H. CH3 "H g) h) Cl
-
Comparative Statement of Financial Position for 2 0 X 4 and 2 0 X 3 , a Statement of income for 2 0 X 4 , and additional information from the accounting records of Olaniyi Industries, are provided...
-
When Patey Pontoons issued 6 % bonds on January 1 , 2 0 2 4 , with a face amount of $ 6 0 0 , 0 0 0 , the market yield for bonds of similar risk and maturity was 7 % . The bonds mature December 3 1 ,...
-
Accountants distinguish between capital and revenue expenditures for some types of transactions. The entries for such transactions can be made to any one of the following accounts: Balance sheet...

Study smarter with the SolutionInn App


