Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions: (a) MgO-25
Question:
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions:
(a) MgO-25 wt% FeO;
(b) MgO-45 wt% FeO;
(c) MgO-65 wt% FeO; and
(d) MgO-80 wt% FeO.
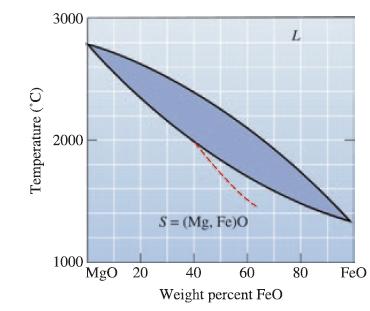
Transcribed Image Text:
Temperature (°C) 3000 2000 1000 MgO 20 S = (Mg,Fe)O 40 60 Weight percent FeO L 80 FeO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a The liquidus temperature 12283 K the solidus temperature 13023 K and the free...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions. (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; (d) MgO-80 wt%...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions: (a) NiO-30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60 mol% MgO; and (d)...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following Al 2 O 3 -Cr 2 O 3 ceramic compositions: (a) Al 2 O 3 -30 wt% Cr 2 O 3 ; (b) Al 2 O 3 -50 wt% Cr 2 O 3 ;...
-
A 4-ft-high, 3-ft-diameter cylindrical water tank whose top is open to the atmosphere is initially filled with water. Now the discharge plug near the bottom of the tank is pulled out, and a water jet...
-
Starting with the algebraic and graphical results of Problems 5 and 6, determine algebraically and determine graphically the effect on YE of an autonomous: (a) Increase in X of 200. (b) Increase in I...
-
Un June 30, 2020, the market interest rate is 7\%. Dellaca Enterprises issues \(\$ 500,000\) of \(8 \%, 20\)-year bonds at 110.625 . The bonds pay interest on June 30 and December 31. Dellaca...
-
In a test of H0: p1 = p2 versus H1: p1 p2, the value of the test statistic is z = 1.21. What do you conclude about the difference p1 p2 at the ???? = 0.05 level of significance?
-
Norr and Caylor established a partnership on January 1, 2019. Norr invested cash of $100,000 and Caylor invested $30,000 in cash and equipment with a book value of $40,000 and fair value of $50,000....
-
Sales MOSS COMPANY Income Statement For Year Ended December 31, 2021 Cost of goods sold Gross profit Operating expenses (excluding depreciation) Depreciation expense Income before taxes Income taxes...
-
Conifer Corp is a sawmill operation in Oregon, that is owned by a major forest products company, but operates independently of headquarters. The mill is divided into six operating departments: boom,...
-
Locate the following points in the Ag-Pd phase diagram and indicate the phases present and their relative amounts: (a) 50 wt% Pd at 1300C; (b) 80 wt% Pd at1425C; and (c) 90 wt% Ag at 1100C....
-
For a Ag-60 wt% Pd alloy determine the (a) Liquidus temperature; (b) Solidus temperature; and (c) Freezing range. Temperature (C) 1600 1500 1400 1300 1200 1100 1000 900 961.93 C 0 10 Ag 20 1555 C 30...
-
If F = 5 kN and = 30, determine the magnitude of the resultant force and its direction, measured counterclockwise from the positive x axis. 4 kN 15 30 6 kN
-
LaVilla is a village in the Italian Alps. Given its enormous popularity among Swiss, German, Austrian, and Italian skiers, all of its beds are always booked in the winter season and there are, on...
-
Using the Monte Carlo setup for the unbalanced one-way error component model considered by Baltagi and Chang (1994), compare the various estimators of the variance components and the regression...
-
Repeat Exercise 13.10 with the specifications below: \[\begin{aligned}A_{\mathrm{p}} & =1.0 \mathrm{~dB} \\A_{\mathrm{r}} & =70 \mathrm{~dB} \\\omega_{\mathrm{p}} & =0.025 \pi \mathrm{rad} /...
-
Plot the pole-zero constellation as well as the magnitude response of the transfer function of Exercise 4.20 for \(M=6,7,8\) and comment on the results. Exercise 4.20 Some FIR filters present a...
-
Using the Monte Carlo setup for the unbalanced two-way error component model considered by Wansbeek and Kapteyn (1989), compare the MSE performance of the variance components and the regression...
-
Match each ratio or percentage with its computation. Ratios or Percentages 1. Net profit margin 2. Inventory turnover ratio 3. Average days to collect receivables 4. Dividend yield ratio 5. Return on...
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
Many lithium salts are hygroscopic (absorb water), but the corresponding salts of the other alkali metals are not. Why are lithium salts different from the others?
-
There are three known xenon fluoride covalent compounds: XeF 2 , XeF 4 , and XeF 6 . In general, the xenon fluoride compounds must be stored in an inert atmosphere, free of oxygen and water. Why is...
-
What are boranes? Why were they once considered as potential fuels for rockets?
-
Influencer marketing on social platforms is integral to marketing campaigns of many brands. For FYiDoctors, it is also vital for the brand. FYIDoctors currently haven't hired any influencers, but it...
-
A conjoint analysis to estimate the partwords of individuals attributed levels of a smartwatch is being designed. There are three levels for the size attributed (40mm, 44mm and 46mm) and three levels...
-
Answer the question below for the bike company, Rad Power Bikes. Marketing Strategy a. The chosen strategy should fit the market and competitive conditions faced by the product and its strategic...

Study smarter with the SolutionInn App


