Determine the liquidus temperature, solidus temperature, and freezing range for the following Al 2 O 3 -Cr
Question:
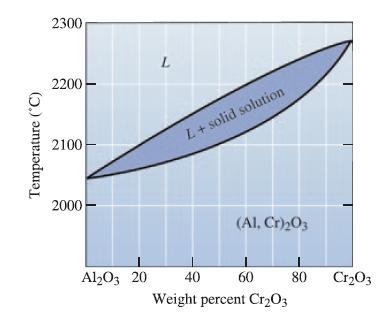
Determine the liquidus temperature, solidus temperature, and freezing range for the following Al2O3-Cr2O3 ceramic compositions:
(a) Al2O3-30 wt% Cr2O3;
(b) Al2O3-50 wt% Cr2O3; and
(c) Al2O3-75 wt% Cr2O3.
Transcribed Image Text:
Temperature (°C) 2300 2200 2100 2000 Al2O3 20 L L + solid solution (Al, Cr)₂03 40 60 Weight percent Cr₂O3 80 Cr₂03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a Liquidus temperature 2160 C Solidus temperat...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions. (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; (d) MgO-80 wt%...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions: (a) NiO-30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60 mol% MgO; and (d)...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; and (d) MgO-80...
-
What is Norway's global health issues and how can they be combated? explain
-
Indicate the expenditure-changing and expenditure-switching policies required to achieve external and internal balance simultaneously for points C 1 , C 4 , C 7 , and C 10 in the following figure...
-
Companies that operate in different industries may have very different financial ratio values. These differences may grow even wider when we compare companies located in different countries Compare...
-
In a set of 12 matched pairs, the mean difference was d = 18 and the standard deviation of the differences was sd = 4. Find the value of the test statistic for testing H0: d = 15 versus H1: d > 15....
-
Charter One Bank owned a fifteen-story commercial building. A fire inspector told Charter that the building's drinking water and fire suppression systems were linked. Without disclosing this...
-
Evaluate the causes and type of data errors in the case senario of Garrison Children Hospital been part of Premier Health care , an academic medical center
-
In each of the following exercises, the assignment is to prepare an office memo. Each assignment contains an assignment memo from the supervising attorney that includes all the available facts of the...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following MgOFeO ceramics at 2000C: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-60 wt%...
-
A NiO-20 mol% MgO ceramic is heated to 2200C. Determine (a) The composition of the solid and liquid phases in both mol% and wt%; (b) The amount of each phase in mol% and wt%; and (c) Assuming that...
-
General Motors stock fell from $32 per share in 2006 to $20 per share during 2008. a. If you bought and then sold 300 shares at these prices, what was your loss? b. Express your loss as a percent of...
-
Consider Mundlak's (1978) augmented regression in (7.35) except now allow for unbalanced panel data. Show that OLS on this augmented regression yields the unbalanced Within estimator for \(\beta\)...
-
An important practice for entrepreneurs and their startups is to involve others in their activities, partly because that is what it takes to conduct a proper feasibility analysis and to do the...
-
Go to the Hubbard OBrien Economics Blog and read the following post titled, How the Effects of the Covid-19 Recession Differed Across Business Sectors and Income Groups,...
-
On 1 July 2019, Weiland Pty Ltd owned several farming vehicles that had cost a total of $155 000. Accumulated depreciation on these vehicles to 1 July 2019 amounted to $73 000. On 30 September 2019,...
-
Acetic acid is to be removed from an aqueous solution by allowing droplets of the acetic acid-water solution to fall through a column of benzene. The droplets initially contain 1.0 weight % of acetic...
-
Company X and Company Y are two giants of the retail industry. Both offer full lines of moderately priced merchandise. In the last fiscal year, annual sales for Company X totaled $53 billion and...
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
When sodium reacts with hydrogen gas, sodium hydride is produced. Is sodium hydride an ionic or a covalent compound? When sodium hydride reacts with water, the equation is: Show that this reaction...
-
Compare the Lewis structures with the molecular orbital view of the bonding in NO, NO + , and NO - . Account for any discrepancies between the two models.
-
Besides the central atom, what are the differences between CO 2 and SiO 2 ?
-
The current time 15 September 15, 2008. The manager of a $500 million short-term loan portfolio would like to use Eurodollar futures to hedge uncertainty of interest receipts. The interest rate on...
-
How do you design a database schema for a simple e-commerce application ? Explain
-
What is a database transaction, and what properties should a transaction have (ACID properties) ? Explain

Study smarter with the SolutionInn App


