Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions: (a) NiO-30
Question:
Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions:
(a) NiO-30 mol% MgO;
(b) NiO-45 mol% MgO;
(c) NiO-60 mol% MgO; and
(d) NiO-85 mol% MgO.
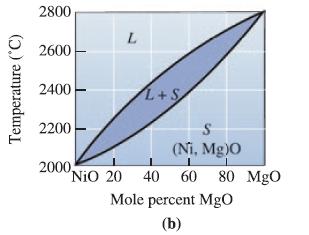
Transcribed Image Text:
Temperature (°C) 2800 2600 2400 2200 L 2000, L+S S (Ni, Mg)0 "NiO 20 40 60 80 MgO Mole percent MgO (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a The liquidus temperature 1515 K and the solidus temperature 1486 K The freezing range will occu...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions. (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; (d) MgO-80 wt%...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following Al 2 O 3 -Cr 2 O 3 ceramic compositions: (a) Al 2 O 3 -30 wt% Cr 2 O 3 ; (b) Al 2 O 3 -50 wt% Cr 2 O 3 ;...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; and (d) MgO-80...
-
How do recruitment and selection practices contribute to high performance in an organization?
-
Starting from point E in Figure 18.8, draw a figure showing how the nation could reach internal and external balance with flexible exchange rates by using an expansionary fiscal rather than an easy...
-
Determine whether the following series converge. k2 -1 k? + 3 k=2 8.
-
How do you plan a mission?
-
Kitchen Gadgets sells a line of high-quality kitchen utensils and gadgets. When customers place orders on the companys Web site or through electronic data interchange (EDI), the system checks to see...
-
Pizza Corporation acquired 75 percent of Slice Corporation's voting common stock on January 1, 20x4, for $351,000, when the fair value of its net identifiable assets was $464,000 and the fair value...
-
Write a program in MIPS assembly language to convert an ASCII number string containing positive and negative integer decimal strings, to an integer. Your program should expect register $a0 to hold...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following Al 2 O 3 - Cr 2 O 3 ceramics at 2150C: (a) Al 2 O 3 -30 wt% Cr 2 O 3 ; (b) Al 2 O 3...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following MgOFeO ceramics at 2000C: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-60 wt%...
-
A consumer products company reported a 5.4 percent increase in sales from 2017 to 2018. Sales in 2017 were $29,600. In 2018, the company reported cost of sales in the amount of $9,107. What was the...
-
An investment group offers their clients advice on creating a diversified investment portfolio. A client asks them to develop a portfolio that includes Investment in a Domestic Growth Fund (a mutual...
-
Ross purchased a new commercial vehicle today for $20,000 inclusive of all tax and dealer fees. He made a $1000 down payment and the balance on the car was financed with using a 5 year loan with a 3%...
-
The risk - free rate r f is 2 . 9 % , and the market return r M is 1 0 . 9 % . ' s beta is 0 . 9 . The company's next four dividends are expected to be $ 0 . 7 2 , $ 0 . 8 1 , $ 0 . 9 , and $ 1 . 0 0...
-
What is the current yield on a zero coupon bond with a remaining life of 7 years, a yield to maturity of 1 5 . 0 % , and a par value of $ 1 0 0 0 ? ( Round to 1 0 0 th of a percent and enter as a...
-
The winner of a lottery is awarded $ 6 , 0 0 0 , 0 0 0 to be paid in annual installments of $ 3 0 0 , 0 0 0 for 2 0 years. Alternatively, the winner can accept a "cash value" one - time payment of $...
-
Describe the proton-coupled 13C NMR spectrum for compounds 1, 3, and 5 in Problem 36, showing relative values (not absolute values) of chemical shifts. 1. CH3CH2CH2Br 3. CH3CH2OCH3 5. CH3CH2COCH3
-
Define relevant costs and discuss: (1) whether all future costs are relevant for decision making and (2) whether variable costs are always relevant and fixed costs are always irrelevant
-
Predict some possible compounds that could form between chlorine and selenium.
-
There are two forms of solid sulfur: rhombic and monoclinic. The stable form of sulfur at 25C is the rhombic form. Upon heating, the rhombic form converts to the monoclinic form, which is the stable...
-
Fluorine reacts with sulfur to form several different covalent compounds. Three of these compounds are SF 2 , SF 4 , and SF 6 . Draw the Lewis structures for these compounds, and predict the...
-
When owning and operating any business, whether it's a caf or a pool cleaning business, it's very important that you know exactly what your income and expenditure is. Is your business making money or...
-
Resources Unlimited Corporation was formed in 1985 through the merger of two natural pipeline companies. The result was the largest gas distribution network in the United States, with 38,000 miles of...
-
Toyota was the world's first automobile manufacturer to produce more than 10 million vehicles per year, which it has done since 2012, when it also reported the production of its 200-millionth...

Study smarter with the SolutionInn App


