How much of a temperature change is required to bring liquid magnesium from solid-liquid equilibrium to vaporliquid
Question:
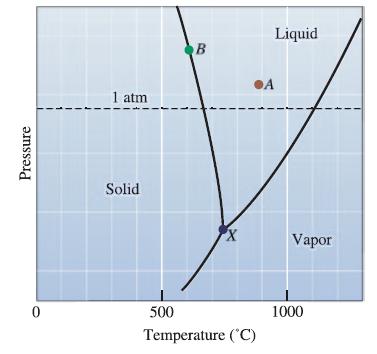
How much of a temperature change is required to bring liquid magnesium from solid-liquid equilibrium to vaporliquid equilibrium at 1 atmosphere?
Transcribed Image Text:
Pressure 0 1 atm Solid B X 500 Temperature (°C) Liquid A Vapor 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Temperature change Power Tempe...View the full answer

Answered By

Willis Omondi
Hi, I'm Willis Omondi, a proficient and professional academic writer. I have been providing high-quality content that best suits my clients and completing their work within the deadline. All my work has been 100% plagiarism-free, according to research from my services, especially in arts subjects and many others
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
How should a monopsonist decide how much of a product to buy? Will it buy more or less than a competitive buyer? Explain briefly.
-
How much of a price premium do you think national brands ought to command over private brands? Justify your position.
-
A government is trying to decide how much of a public good to provide. The willingness-to-pay curves for each of its two citizens are as given in the diagram. The marginal cost curve for the public...
-
You are the manager of a local coffee shop. There are two types of customers in your market, coffee addicts, and casual drinkers. Because coffee addicts buy large amounts of coffee, they are more...
-
Starting with the exchange rate of R = $2/1, draw a figure showing the exchange rate under a crawling peg system with the nation appreciating its currency by 1 percent at the end of each month for...
-
(a) Show that for an analytic signal representation of a real-valued, narrowband random process, the autocorrelation function of the resulting complex process \(\mathbf{U}(t)\) (assumed wide-sense...
-
University endowments are financial assets that are donated by supporters to be used to provide income to universities. There is a large discrepancy in the size of university endowments. The...
-
Flexible-budget and sales volume variances. Marron, Inc. produces the basic fillings used in many popular frozen desserts and treatsvanilla and chocolate ice creams, puddings, meringues, and fudge....
-
How does a community service review and revise their work practices following feedback from the community? How would you revise the work delivered if social and cultural issues were not being...
-
The accounting records of Blue Bird Co. showed the following balances at January 1, Year 2: Cash..............................................................................................$30,000...
-
How can solid solutions form in ceramic systems?
-
Do we need 100% solid solubility to form a solid solution of one material in another?
-
Write an algorithm to determine whether the rules of an arbitrary attribute grammar are noncircular. (Your algorithm will require exponential time in the worst case [JOR75].)
-
Greetings to you my beloved tutors and/or knowledge provider. Kindly assist with the below question. You may give me an overview with reference to any journal articles, books, case law, etc. The...
-
Whistleblowing policies are designed to encourage employees, suppliers, customers, and other stakeholders to report suspected misconduct or fraudulent activities within an organization. These...
-
In the digital age, cybersecurity auditing and risk management have become essential components of organizational security strategies. Cybersecurity auditing involves the systematic assessment of an...
-
How are environmental laws impacted by Public Use?
-
Suppose a non-recognition transaction occurs eight months prior to a liquidation being adopted. However, the acquisition was not motivated by a loss recognition purpose. Section 351 provides...
-
The radioactive potassium-40 isotope decays to argon-40 with a half-life of 1.2 109 yr. (a) Write a balanced equation for the reaction. (b) A sample of moon rock is found to contain 18 percent...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Which of the following statement(s) is(are) true? a. A radioactive nuclide that decays from 2.00 10 21 atoms to 5.0 10 20 atoms in 16 minutes has a half-life of 8.0 minutes. b. Nuclides with large...
-
A certain radioactive nuclide has a half-life of 3.00 hours. a. Calculate the rate constant in s -1 for this nuclide. b. Calculate the decay rate in decays/s for 1.000 mole of this nuclide.
-
Complete the following table with the nuclear particle that is produced in each nuclear reaction. Initial Nuclide 23 Pu 94 214Pb 82 60, 27 99- 43 Tc 93Np Product Nuclide 2351 92 214p 60 Ni 28 44Ru...
-
Solve the initial value problem. dy =3e3t sin (e3t-27), y(In 3)=0 dt The solution is y =
-
Solve by factoring. 5x2-16x+12=0
-
Find f-1 (x) for f (x) = 15 + 12x.

Study smarter with the SolutionInn App


