In the presence of nitric acid, UO 2+ undergoes a redox process. It is converted to UO
Question:
In the presence of nitric acid, UO2+ undergoes a redox process. It is converted to UO22+ and nitric oxide (NO) gas is produced according to the following unbalanced equation:
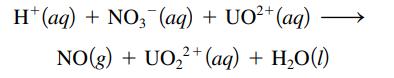
If 2.55 × 102 mL NO(g) is isolated at 29οC and 1.5 atm, what amount (moles) of UO2+ was used in the reaction?
Transcribed Image Text:
H+ (aq) + NO3(aq) + UO²+ (aq) NO(g) + UO₂²+ (aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Amount moles of ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
If 2-methylpropane is brominated at 125C in the presence of light, what percent of the product will be 2-bromo-2-methylpropane? Compare your answer with the percent given in Problem 4 for...
-
In the presence of the enzyme aconitase, the double bond of aconitic acid undergoes hydration. The reaction is reversible, and the following equilibrium is established:
-
At 500 K in the presence of a copper surface, ethanol decomposes according to the equation C2H5OH(g) CH3CHO(g) + H2(g) The pressure of C2H5OH was measured as a function of time, and the following...
-
Why do we need Normalization? How many forms of Normalization exist? What is the minimum normal form required? Describe each normal form List Normalization conversion process
-
In Shelanu v. Three Print, the Ontario Court of Appeal held that franchisors owe a duty of good faith to franchisees. What factors led the court to this conclusion? In what other contractual...
-
On May 15, 19X2, you are engaged to make an audit of Boro Corporation, whose records you have not previously audited. Your client is a retail coal dealer with a fiscal year ending May 31. Perpetual...
-
What are the ethical obligations in using social media sites in litigation?
-
The following data pertain to the Aquarius Hotel Supply Company for the year just ended. Budgeted sales revenue.................................................................$945,000 Budgeted...
-
SERVICE AT HOTEL ALORE. Last month, Julian had been asked to attend a three-day conference in a city five four hours from his home. He and his wife had decided to make it a weekend getaway for the...
-
Required Tasks: 1. Prepare a descriptive analysis of the data using charts, graphs, and numerical measures. 2. Construct and interpret a 95% confidence interval estimate for the mean weight for male...
-
We state that the ideal gas law tends to hold best at low pressures and high temperatures. Show how the van der Waals equation simplifies to the ideal gas law under these conditions.
-
A large flask with a volume of 936 mL is evacuated and found to have a mass of 134.66 g. It is then filled to a pressure of 0.967 atm at 31 C with a gas of unknown molar mass and then reweighed to...
-
The combustion of 1.00 mol liquid octane (C 8 H 18 ), a component of gasoline, in excess oxygen is exothermic, producing 5.46 10 3 kJ of heat. (a) Write the thermochemical equation for this...
-
Using real-world examples, discuss the possible relationship between economic growth and economic development.
-
Murchison Technologies, Inc. recently developed a patient-billing software system that it markets to physicians and dentists. Jim Archer and Janice Johnson founded the company in Austin, Texas five...
-
Interview at least ten people and ask the following questions: What are your auto liability insurance limits and deductibles? If you own your own home, do you have an all-risk or named perils...
-
Lee Chiew is borrowing \($170\),000 from Asia Bank USA to open Asian Health Foods in Virginia. She agrees to repay the loan at 10 percent compounded annually. Since she expects it to take a few years...
-
A series of 10 end-of-year deposits is made that begins with $7,000 at the end of year 1 and decreases at the rate of $300 per year with 10 percent interest. a. What amount could be withdrawn at t =...
-
The controller of Hall Industries has collected the following monthly expense data for use in analyzing the cost behavior of maintenance costs. Instructions (a) Determine the fixed- and variable-cost...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
A compound with molecular formula C 10 H 10 O 4 produces a 1 H NMR spectrum that exhibits only two signals, both singlets. One signal appears at 3.9 ppm with a relative integration value of 79. The...
-
For each of the following compounds, predict the number of signals and location of each signal in a 13 C NMR spectrum: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) H.
-
Compare the following two constitutional isomers. The 13 C NMR spectrum of the first compound exhibits five signals, while the second compound exhibits six signals. Explain. .
-
If the high temperature steam has a temperature of 2 9 7 . 7 K and the low temperature reservoir has a temperature of 1 4 7 . 8 K what is the carnot efficiency of this process
-
Discuss the innovative idea/project addressing the problem below by the following (a) potential impact - (b) novelty and innovation- (c) feasibility- (d) sustainability- Reference: i. Potential...
-
Marianne's duplex sold at a foreclosure auction for $280,000. At the time of foreclosure, she had not made any payments to the bank for 11 months ($2,400 each). The outstanding balance of her...

Study smarter with the SolutionInn App


