The rate constant for the gas-phase decomposition of N 2 O 5 , has the following temperature
Question:
The rate constant for the gas-phase decomposition of N2O5,
![]()
has the following temperature dependence:
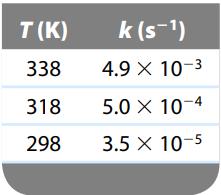
Make the appropriate graph using these data, and determine the activation energy for this reaction.
Transcribed Image Text:
N₂O, (g) 2NO₂(g) + O₂(g)
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The temperature dependence of the rate constant for a reaction is tabulated as follows: Temperature (K) k (M-1 s-1) 600................................0.028 650................................0.22...
-
The rate constant for the gas- phase decomposition of N2O5, N2O5 2NO2 + 1/2O2 has the following temperature dependence: Make the appropriate graph using these data, and deter-mine the activation...
-
A) For the gas phase decomposition of hydrogen iodide at 700 K 2 HIH2 + I2 the average rate of disappearance of HI over the time period from t = 0 s to t = 1446 s is found to be 5.6710-4 M s-1. The...
-
Suppose that, in an attempt to raise more revenue, Nowhere State University (NSU) increases its tuition. Will this necessarily result in more revenue? Under what conditions will revenue (a) rise, (b)...
-
What is viral marketing? How do marketers use brand ambassador or brand evangelists?
-
A particularly long traffic light on your morning commute is green 20% of the time that you approach it. Assume that each morning represents an independent trial. (a) Over five mornings, what is the...
-
Water for a Pelton wheel turbine flows from the headwater and through the penstock as shown in Fig. P12.54. The effective friction factor for the penstock, control valves, and the like is 0.032 , and...
-
Linda Day George Company had bonds outstanding with a maturity value of $300,000. On April 30, 2014, when these bonds had an unamortized discount of $10,000, they were called in at 104. To pay for...
-
Analytically re-calculate the force R acting on point C. It will no longer be 687.5 N as previously found and listed in the slides 800 N 500 N A 55 40 40 D B 80 60 R C
-
Suppose you executed the following trade (strategy) on NPR Ltd using two exercise prices: Long 1 NPR call (X=R50) @ R8.40 Short 2 NPR calls (X =R60) @ R3.34 each All options have identical expiration...
-
DDT (molar mass = 354.49 g/mol) was a widely used insecticide that was banned from use in the United States in 1973. This ban was brought about due to the persistence of DDT in many different...
-
One reason suggested for the instability of long chains of silicon atoms is that the decomposition involves the transition state shown below: The activation energy for such a process is 210 kJ/mol,...
-
Information related to Mingenback Ltd. for 2020 is summarized below. Total credit sales........................................................ 2,500,000 Accounts receivable at December...
-
With regard to a group life insurance policy. What is the purpose of medical evidence of insurability?
-
What is the difference between training and career development? Of these two concepts, which is more likely to increase retention in an organization? Why?
-
List the aspects of Aboriginal and/or Torres Strait Islander Peoples ways of learning the mathematics, music, art, and science.
-
The facts of the case are simple: A dispute in the shop at an auto parts manufacturing plant in Hamilton, Iowa, ended when one worker murdered another. At 2:00 p.m. police responded to a report of a...
-
What are there penalties for companies like Starbucks that refuse employees the right to unionize? Explain briefly
-
The running engine of an automobile produces carbon monoxide (CO), a toxic gas, at the rate of about 188 g CO per hour. A car is left idling in a poorly ventilated garage that is 6.0 m long, 4.0 m...
-
Solve the relation Exz:Solve therelation ne %3D
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
make a scenario for each of the following Gesture communication Facial expression communication Oral communication Written communication discuss briefly each scenario
-
Problem 7-30 Holding Period Yield (LO2) The YTM on a bond is the interest rate you earn on your investment if interest rates don't change. If you actually sell the bond before it matures, your...
-
According to theknot.com, online dating is the most common way people meet their spouses today. This claim was based on a survey of 1,400 randomly selected recently married couples, with 266...

Study smarter with the SolutionInn App


