What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise
Question:
What amount (moles) of compound is present in 1.00 g of each of the compounds in Exercise 53?
Data in Exercise 53?
Calculate the molar mass of the following substances.
a. 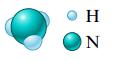
b. 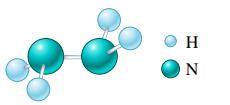
c. (NH4)2Cr2O7
Transcribed Image Text:
Η N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
c NH42Cr2O7 To calculate the molar mass of NH42Cr2O7 we need to sum up the atomic mas...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Calculate the molar mass of the following substances: (a) Li2 CO3, (b) CS2, (c) CHCl3 (chloroform), (d) C6H8O6 (ascorbic acid, or vitamin C), (e) KNO3, (f) Mg3N2.
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 54? Data In Exercise 54? Calculate the molar mass of the following substances. a. b. Ca 3 (PO 4 ) 2 c. Na 2 HPO 4 0
-
What mass of compound is present in 5.00 moles of each of the compounds in Exercise 53? Data in Exercise 53? Calculate the molar mass of the following substances. a. b. c. (NH 4 ) 2 Cr 2 O 7 OH N
-
Find the x-values where the following do not have derivatives. + -6 + y 8 0 6 X
-
Use the Holt Winters no trend model to find the best model to find forecasts for the next 12 months in the Excel file Housing Starts.
-
A 1-mm-thick layer of water on an electrically heated plate is maintained at a temperature of \(T_{w}=340 \mathrm{~K}\), as dry air at \(T_{\infty}=300 \mathrm{~K}\) flows over the surface of the...
-
An incompressible fluid flows outward through a blower as indicated in Fig. P5.81. The shaft torque involved, \(T_{\text {shaft }}\), is estimated with the following relationship: \[ T_{\text {shaft...
-
Allen Shonton recently opened his own accounting firm on April 1, which he operates as a corporation. The name of the new entity is Allen Shonton, CPA. Shonton experienced the following events during...
-
. What is a non-political socially-appropriate response to someone who is exhibiting prejudice? What are the responsibilities of a social psychology researcher when the researcher encounters...
-
1. The following figure shows one possible unrooted tree with 5 taxa. ABC / D E How many rooted trees can be derived from this tree? Illustrate them all. [10]
-
What is the mass of the product? a. Less than 10 g b. Between 20 and 100 g c. Between 100 and 120 g d. Exactly 120 g e. More than 120 g
-
Fig. 3.5 illustrates a schematic diagram of a combustion device used to analyze organic compounds. Given that a certain amount of a compound containing carbon, hydrogen, and oxygen is combusted in...
-
Briefly describe the worksheet method of analyzing the information for a companys statement of cash flows. (Do not list the steps in preparation.)
-
According to the NCAA, most athletic departments appear to lose money. Coakley says it's because Group of answer choices . they only calculate generated revenues and subtract total expenses, which...
-
Mr. Schminkie is the sole proprietor of a manufacturing business. He was advised to incorporate a company. Mr. Schminkie could transfer all his assets to the newly formed company and take back debt...
-
Use the horizontal model, or write the journal entries, to show the effect of selling each of the three assets for their respective estimated market values. Partial - year depreciation adjustments...
-
7. What methods of Map can be implemented in O(1) time with a good hash function and a properly-sized hash table? What methods of Set? Could we implement List with a hash table? Explain.
-
Required information LO 4-1: Understand the basic approach in activity-based costing and how it differs from conventional costing. Skip to question In this Learning Objective, we will learn about the...
-
Calculate the number of spheres that would be found within a simple cubic, a body-centered cubic, and a face-centered cubic cell. Assume that the spheres are the same?
-
3M Company reports the following financial statement amounts in its 10-K report: a. Compute the receivables, inventory, and PPE turnover ratios for both 2018 and 2017. (Receivables turnover and...
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Show preparation of these alcohols fromalkenes. b) c)
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
Milligan's Backyard Storage Kits, a mail order company, sells a variety of backyard storage unit kits and landscaping decorations to its customers. Although the company makes a profit, David...
-
1. If you think about then Guest Services Essentials class, what do you believe to be the biggest change in your customer service practice? 2. Name and describe two customer service techniques to...
-
Packaging Waste & Recycling UK, 2013('000 tonnes) 2,260 Total Packaging Waste Total Recycled 4,000 3,868 3,459 3,000 2,339 2,000 1,639 1,029 1,000 806 714 462 436 23 0 0 Metal Glass Wood Paper...

Study smarter with the SolutionInn App


