Determine the total apparent specific heat at constant pressure (c p,mix in kJ/kg K) for a
Question:
Determine the total apparent specific heat at constant pressure (cp,mix in kJ/kg · K) for a fuel–air reactant mixture containing 1 kmol CH4, 2.5 kmol O2, and 9.4 kmol N2 at 500 K and 1 atm. Use specific heat values for each species from Table E.1.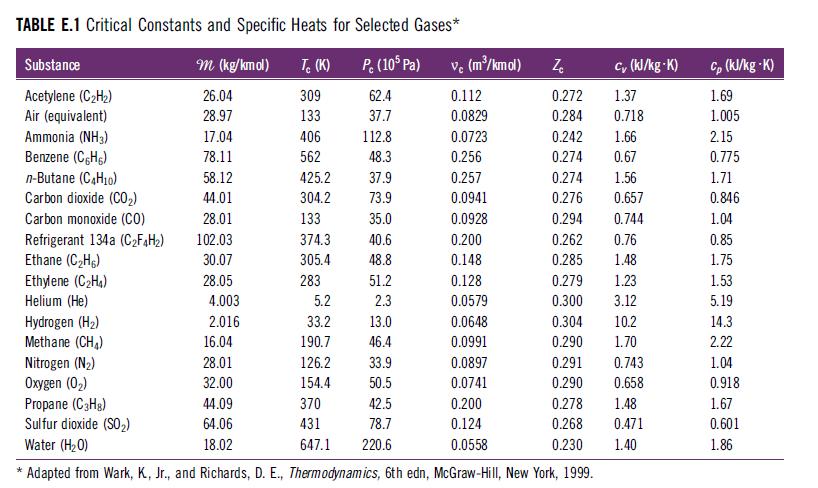
Transcribed Image Text:
TABLE E.1 Critical Constants and Specific Heats for Selected Gases* P. (105 Pa) 62.4 37.7 Substance Acetylene (C₂H₂) Air (equivalent) Ammonia (NH3) Benzene (CH) n-Butane (C4H10) Carbon dioxide (CO₂) Carbon monoxide (CO) * Refrigerant 134a (C₂F4H₂) Ethane (C₂Hg) Ethylene (C₂H4) Helium (He) m (kg/kmol) 26.04 28.97 17.04 78.11 58.12 44.01 28.01 102.03 30.07 28.05 4.003 2.016 Tc (K) 309 133 406 562 425.2 304.2 133 374.3 305.4 283 5.2 33.2 190.7 126.2 154.4 370 431 647.1 112.8 48.3 37.9 73.9 35.0 40.6 48.8 51.2 2.3 13.0 46.4 33.9 50.5 42.5 78.7 220.6 vc (m³/kmol) 0.112 0.0829 0.0723 0.256 0.257 0.0941 0.0928 0.200 0.148 0.128 0.0579 cy (kJ/kg-K) 1.37 0.718 0.242 1.66 0.274 0.67 0.274 1.56 0.276 0.657 0.294 0.744 0.262 0.76 0.285 1.48 Zc 0.272 0.284 0.279 1.23 0.300 3.12 Hydrogen (H₂) 0.0648 Methane (CH₂) 16.04 0.0991 Nitrogen (N₂) 28.01 0.0897 Oxygen (0₂) 32.00 0.0741 Propane (C3H8) 44.09 0.200 Sulfur dioxide (SO₂) 64.06 0.124 Water (H₂O) 18.02 0.0558 Adapted from Wark, K, Jr., and Richards, D. E., Thermodynamics, 6th edn, McGraw-Hill, New York, 1999. 0.304 0.290 0.291 0.290 10.2 1.70 0.743 0.658 0.278 1.48 0.268 0.471 0.230 1.40 Cp (kJ/kg-K) 1.69 1.005 2.15 0.775 1.71 0.846 1.04 0.85 1.75 1.53 5.19 14.3 2.22 1.04 0.918 1.67 0.601 1.86
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To determine the total apparent specific heat at constant pressure for the given fuelair mixture we ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Determine the total apparent specific heat at constant volume (c v,mix in kJ/kgK) for a fuelair reactant mixture containing 1 kmol CH 4 , 2.5 kmol O 2 , and 9.4 kmol N 2 at 500 K and 1 atm. What heat...
-
A mixture of ideal gases contains 0.5 kmol of CO 2 , 2 kmol of O 2 , and 7 kmol of N 2 at 700 K. Determine the following quantities: A. The mole fraction of each constituent in the mixture. B. The...
-
Complete the following questions. Submit journal entries in an Excel Template, linked at the bottom of this page, and any written segments in Excels comments function. Do not submit two separate...
-
Format only cells with values greater than maxim 4 tab and select Red color from the pallet (bottom ro Format only cells with values less than minimum and select Yellow color from the pallet (bottom...
-
Obtain IRS Revenue Ruling 59 60 through the IRS web site. Read this Ruling and prepare a memo to your professor explaining how each of its eight valuation factors fits into the theory of business...
-
You receive a check drawn on another bank and deposit it into your checking account. Even though this is a demand deposit the funds are not immediately available for your use. Why? Would your answer...
-
Refer to the information in Exercise 17-7 to answer the following requirements. Required 1. Using ABC, compute the overhead cost per unit for each product line. 2. Determine the total cost per unit...
-
A payroll summary for Mark Consulting Company, owned by Mark Fronke, for the quarter ending June 30, 2016, appears on page 360. The firm made the required tax deposits as follows: a. For April taxes,...
-
A bank in Toronto charges 2.2% commission to buy and sell currencies. Assume that the current exchange rate is US$1 = C$1.1153. a. How many Canadian dollars will you have to pay to purchase US$4,500?...
-
Complete the following table: Assets Liabilities + Owner's Equity . $17,500 ? + $11,500 b. ? $5,000 + $60,000 C. $15,000 $12,000 +
-
A 1-m 3 tank contains nitrogen at 30C and 500 kPa. In an isothermal process, CO 2 is forced into this tank until the pressure is 1000 kPa. What is the mass (kg) of each gas present at the end of this...
-
A 17.3-liter tank contains a mixture of argon, helium, and nitrogen at 298 K. The argon and helium mole fractions are 0.12 and 0.35, respectively. If the partial pressure of the nitrogen is 0.8 atm,...
-
Propylene gas at I27oC and 38 bar is throttled in a steady-state flow process to 1 bur, where it may be assumed to be an ideal gas. Estimate the final temperature of the propylene and its entropy...
-
5. Polanti Company's sales and inventory data for the second quarter of the year: Product T sales April May June 320,000 290,000 265,000 Product X sales 130,000 154,000 198,000 Anticipated beginning...
-
1. Calculate the alpha, tracking error and information ratio for the following portfolio Period Portfolio Benchmark 1 16.25% 15.89% 2 22.18% 23.62% 3 -4.78% -4.82% 4 9.56% 9.65% 5 10.14% 10.11% 67...
-
While you are having a discussion with a colleague, she mentions that she believes it would be much easier and would have a minimal effect on the financial statements if we eliminated all adjusting...
-
You are considering moving some of your operations to a state/country that you estimate would reduce your overall tax rate 0.077 (770 basis points) Tax burden 0.77 Interest burden 0.85 Operating...
-
Star Rock is a Canadian micro brewery that is operating at full capacity. The company would like to have an international presence (China) and has seen that tourists and expats show positive buying...
-
Rickys Piano Rebuilding Company has been operating for one year. On January 1, at the start of its second year, its income statement accounts had zero balances and its balance sheet account balances...
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Determine the value of the stoichiometric coefficients for the combustion of coal in oxygen given by the stoichiometric equation: CHN 0:01 O 0:1 S 0:05 + aO 2 = bCO 2 + cH 2 O + dN 2 + eSO 2
-
Determine the value of the stoichiometric coefficients for the combustion of natural gas in air: CH 4 + a(O 2 + 3:76N 2 ) = bCO 2 + cH 2 O + dN 2
-
Using the stoichiometric coefficients you found in exercise 9, determine the molar air-to-fuel ratio (A/F) molar for the combustion of natural gas in air. (A: (A/F) molar = 9.52 kmol of air/kmol of...
-
The purpose of this assignment is to determine if the database performance can be improved by monitoring response times for frequently used queries and determining if the queries were written...
-
Study the index definitions in sh_idx.sql. These indexes have already been created in SH2. Whatever indexes you decide to create for this task should be the result of your own research and thinking...
-
What is an interesting order in terms of query optimization in databases? Can you give one or two examples and explain how they can be useful in getting a better physical query plan?

Study smarter with the SolutionInn App


