In Table C.2, at what reference temperature and pressure is the entropy zero? TABLE C.2 Thermodynamic Properties
Question:
In Table C.2, at what reference temperature and pressure is the entropy zero?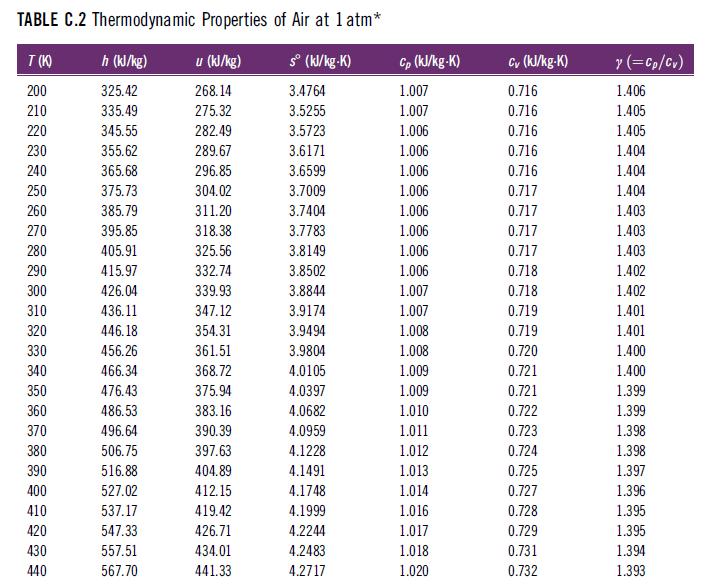
Transcribed Image Text:
TABLE C.2 Thermodynamic Properties of Air at 1 atm* h (kJ/kg) u (kJ/kg) sº (kJ/kg-K) 325.42 268.14 3.4764 335.49 275.32 3.5255 345.55 282.49 3.5723 355.62 289.67 3.6171 365.68 296.85 3.6599 375.73 304.02 3.7009 385.79 311.20 3.7404 395.85 318.38 3.7783 405.91 325.56 3.8149 415.97 332.74 3.8502 426.04 339.93 3.8844 436.11 347.12 3.9174 446.18 354.31 3.9494 456.26 361.51 3.9804 466.34 368.72 4.0105 476.43 375.94 4.0397 486.53 383.16 4.0682 496.64 4.0959 506.75 4.1228 516.88 4.1491 527.02 4.1748 537.17 4.1999 547.33 4.2244 557.51 4.2483 567.70 4.2717 T(K) 200 210 220 230 240 250 260 270 280 290 300 310 320 330 340 350 360 370 380 390 400 410 420 430 440 390.39 397.63 404.89 412.15 419.42 426.71 434.01 441.33 Cp (kJ/kg-K) 1.007 1.007 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.006 1.007 1.007 1.008 1.008 1.009 1.009 1.010 1.011 1.012 1.013 1.014 1.016 1.017 1.018 1.020 Cv (kJ/kg-K) 0.716 0.716 0.716 0.716 0.716 0.717 0.717 0.717 0.717 0.718 0.718 0.719 0.719 0.720 0.721 0.721 0.722 0.723 0.724 0.725 0.727 0.728 0.729 0.731 0.732 y (=Cp/Cv) 1.406 1.405 1.405 1.404 1.404 1.404 1.403 1.403 1.403 1.402 1.402 1.401 1.401 1.400 1.400 1.399 1.399 1.398 1.398 1.397 1.396 1.395 1.395 1.394 1.393
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Table C2 lists the specific enthalpy h and specific entropy s of various pure substances at differen...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The following table presents the solubilities of several gases in water at 25 C under a total pressure of gas and water vapor of 1 atm. (a) What volume of CH 4 (g) under standard conditions of...
-
Euler's original article about the Konigsberg Bridge Problem, which is dated 1736, presents a second similar problem with two islands, four rivers flowing around them, and 15 bridges connecting...
-
The molar volume of a certain solid is 142.0 cm-1 mol-1 at 1.00 atm and 427.15 K, its melting temperature. The molar volume of the liquid at this temperature and pressure is 152.6 cm-1 mol-1. At 1.2...
-
Suppose that we want to know the mass of gasoline in an automobile's gas tank. The tank has a volume of 70 L, and a handbook of fluid properties states that the density of gasoline is 736 kg/m. (1000...
-
Reconsider the valuation of the retail clothing chain presented in the previous problem. Use the following benefit stream information for this purpose. You have selected the capitalization of...
-
Find the steady-state current in the RLC-circuit in Fig. 61 for the given data. Show the details of your work. R = 2 , L = 1 H, C = 1/20 F, E = 157 sin 3t V L E(t) = E, sin ot Fig. 61. RLC-circuit
-
How to oppose a motion for summary judgment?
-
The Sherill Utility District was recently established. Its balance sheet, after one year, is presented below. Note the following additional information: ¢ The general fund received all of its...
-
Option Closing Price Strike Price Call-Last Quote September Put-Last Quote September 1 41.50 45.50 4.00 4.40 2 41.50 37.50 6.00 2.40 3 41.50 47.50 3.20 5.20 If you could exercise the options listed...
-
A coach must select two players to serve as captains. He wants to randomly select two players to be the captains. Obtain a simple random sample of size 2 from the following list: Mady, Breanne, Evin,...
-
A 17.3-liter tank contains a mixture of argon, helium, and nitrogen at 298 K. The argon and helium mole fractions are 0.12 and 0.35, respectively. If the partial pressure of the nitrogen is 0.8 atm,...
-
A 3-ft 3 rigid vessel contains a 5050 mixture of N 2 and CO (by volume). Determine the mass of each component for T = 65 F and P = 30 psia.
-
Determine whether the sequence is a Fibonacci-type sequence (each term is the sum of the two preceding terms). If it is, determine the next two terms of the sequence. 1, 4, 9, 16, 25, 36, . . .
-
Provide benchmarks against which you can measure yourself. Your ability, for example, to achieve what your culture defines as success contributes to a positive self-concept; your failure to achieve...
-
When managers use the I/O model, why do they consider industry characteristics such as economies of scale and market friction in their decision-making? It highlights unique resources that are...
-
What are the different changes that will dramatically occur during adolescence, Briefly explain each. 2) How do quality relationships influence positive outcomes?
-
(i) In what way can the Luxemburg approach help us to understand the process of globalization in the contemporary economy. (ii) What other arguments from the perspective of the critique of Marxist...
-
How well can we measure the asymptotic frequency? I have run a number of computer simulations in which a 'success' occurred with probability p. It would be no fun if I told you the value of p, but I...
-
A single carousel storage system has an oval rail loop that is = 30 ft long and 3 ft wide. Sixty carriers are equally spaced around the oval. Suspended from each carrier are 5 bins. Each bin has a...
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
Determine the mass air-to-fuel ratio (A/F)mass for the combustion of isooctane C 8 H 18 in air. Its stoichiometric equation is C 8 H 18 + 12:5(O 2 + 3:76N 2 ) = 8CO 2 + 9H 2 O + 47:0N 2
-
Assume one kg of isooctane produces 45,500 kJ of energy. How much carbon dioxide is released in obtaining a kJ of energy from the combustion of isooctane in air? (A: 6.79 10 5 kg CO 2 /kJ) Use...
-
Assume a kg of hydrogen in combustion produces 1.20 10 5 kJ of thermal energy. How much carbon dioxide is released in obtaining a kJ of energy from the combustion of hydrogen in air? Use engineering...
-
As with any counselling issue, IPV can be a stressful area in which to work as a crisis counsellor. Some dynamics of violence, such as life-and-death situations or innocent individuals being hurt,...
-
The Bliss's major presence is in the luxury and upper midscale segment, for which they had invested $ 20 billion in building premium hotel rooms and maintaining them was also an expensive affair....
-
This question is about the application of linear programming (LP). Part (b) is a continuation of Part (a), and Part (c) is not related to Parts (a) and (b). (a) Mary's breakfast is a mix of two...

Study smarter with the SolutionInn App


