Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it
Question:
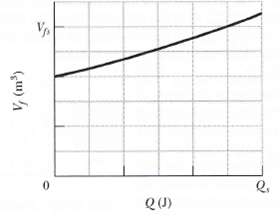
Suppose 0.825 mol of an ideal gas undergoes an isothermal expansion as energy is added to it as heat Q. It Figure 19-20 shows the final volume V1versus Q, what is the gas temperature? The scale of the vertical axis is set by Vfs = 0.30 m3, and the scale of the horizontal axis is set by Qs =1200J.
Transcribed Image Text:
Q()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Using Eq 1914 we note that since it is an isothermal process invol...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A quantity of an ideal gas undergoes an isothermal expansion at 20 oC and does 3.0 x 103 J of work on its surroundings in the process. (a) Will the entropy of the gas (1) increase, (2) remain the...
-
Suppose 4.00 mol of an ideal gas undergoes a reversible isothermal expansion from volume V1 to volume V2 = 2.00 V1 at temperature T = 400 K. Find (a) The work done by the gas and (b) The entropy...
-
The temperature of 0.150 mol of an ideal gas is held constant at 77.0oC while its volume is reduced to 25.0% of its initial volume. The initial pressure of the gas is 1.25 atm. (a) Determine the work...
-
The following MINITAB output exhibits the cumulative distribution function curves of three lognormal distributions. Their mean and variance, respectively, are (1, .5), (1, 1), and (1, 2). Compare the...
-
In 2014, John opened an investment account with Randy Hansen, who held himself out to the public as an investment adviser and securities broker. John contributed $200,000 to the account in 2014. John...
-
The numbers of workers and the numbers of workers in unions are shown in Table 35 for various states. a. Identify the individuals. b. Identify the variables. c. Identify the data for each variable....
-
A temperature sensor of \(10.5-\mathrm{mm}\) diameter experiences cross flow of water with a free stream temperature of \(80^{\circ} \mathrm{C}\) and variable velocity. Derive an expression for the...
-
Tonys Textiles uses a predetermined factory overhead rate based on machine-hours. For May, Tonys budgeted overhead was $210,000 based on a budgeted volume of 35,000 machine-hours. Actual overhead...
-
The controller of Rather Production has collected the following monthly expense data for analyzing the cost behavior of electricity costs. Total Electricity Costs Total Machine Hours January $2,500...
-
A rural clinic hires its staff from nearby cities and towns on a part-time basis. The clinic attempts to have a general practitioner (GP), a nurse, and an internist on duty during at least a portion...
-
Water bottle in a hot car in the American Southwest, the temperature in a closed car parked in sunlight during the summer can be high enough to burn flesh. Suppose a bottle of water at a refrigerator...
-
In the temperature range 310 K to 330 K, the pressure p of a certain non ideal gas is related to volume V and temperature T by how much work is done by the gas if its temperature is raised from 315 K...
-
Determine the number of integer solutions to x1 + x2 + x3 + x4 = 19 where - 5 < x1 < 10 for all 1 i 4.
-
QUESTION 3 (15 MARKS) You are now asked to consider a world with two people, Jane and Jack. Jack is a farmer. Jane owns the land on which Jack farms and does not produce any output herself. There is...
-
You sold product to a customer and received payment at the time of sale. Now you need to record the sales in QuickBooks. Which form would you use?
-
Sport in Society (Coakley) Briefly explain what concerns scholars who look at sport through the prism of conflict theory have. Coakley says students often are resistant to thinking about conflict...
-
How do advanced cost-benefit analysis frameworks and probabilistic decision-making models guide strategic resource allocation decisions, balancing short-term efficiency gains with long-term...
-
NEGOTIATION 1.There is a client complaining about the product they have received. Your customer told you "The item that you send to me has a lot of damages and it is not what I ordered!"Now, the...
-
In Michael Johnsons world-record 400 m sprint, he ran the first 100 m in 11.20 s; then he reached the 200 m mark after a total time of 21.32 s had elapsed, reached the 300 m mark after 31.76 s, and...
-
Question 2 For an n x n matrix A = form) via (aij)
-
If you deposit $2,000 in a 5-year certificate of deposit at 5.2%, how much will it be worth in five years?
-
At the surface of Jupiter's moon 10, the acceleration due to gravity is g = 1.81 mfs2. A watermelon weighs 44.0 N at the surface of the earth. (a) What is the watermelon's mass on the earth's...
-
An astronaut's pack weighs 17.5 N when she is on earth but only 3.24 N when she is at the surface of an asteroid. (a) What is the acceleration due to gravity on this asteroid? (b) What is the mass of...
-
World-class sprinters can accelerate out of the starting blocks with an acceleration that is nearly horizontal and has magnitude 15 m/s2. How much horizontal force must a 55-kg sprinter exert on the...
-
A project costs $80 MM, with 25% of the investment int in year 1 and the balance in year 2. The plant operates atates at 50% capacity in year 3 and then full capacity for the next next 20 years. The...
-
What is organizational Behaviour the study of? How is social sciences related to organizational behavior? What is the difference between social science and organizational behavior? What is the...
-
Describe the nature of organization change, including forces for change and planned versus reactive change

Study smarter with the SolutionInn App


