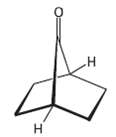
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen
Question:
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen atoms. Explain.
Transcribed Image Text:
エ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
The first step of an aldol condensation is enolate for...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following compound is not aromatic even though it has 4n + 2 electrons in a continuous cyclic array. Explain why this compound is not aromatic.
-
The trial balance of Dominic Company shown below does not balance. Your review of the ledger reveals that each account has a normal balance. You also discover the following errors. 1. The totals of...
-
The trial balance of Zoop Co. shown below does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the following...
-
1. What responsibility does an organization have to ensure that its suppliers and business partners behave ethically? To whom is this responsibility owed? 2. How can an organization monitor the...
-
In the United States, lawyers in negligence cases are usually paid a contingency fee equal to roughly 30 percent of the total award. Lawyers in other types of cases are often paid on an hourly basis....
-
At 20C (approximately room temperature) the average velocity of N2 molecules in air is 1050 mph. (a) What is the average speed in m/s? (b) What is the kinetic energy (in J) of an N2 molecule moving...
-
What are the risks of offshoring work/jobs to other countries, for example, to China or India?
-
In its income statement for the year ended December 31, 2014, Michael Company reported the following condensed data. Instructions (a) Prepare a multiple-step income statement. (b) Prepare a...
-
How do intrinsic and extrinsic motivators impact employee engagement, and what role does organizational design play in fostering a motivated workforce ?
-
You, CPA, work as a consultant on various engagements. Your client, Over The Edge Ltd. (OTE), has grown from a small custom snowboard manufacturer servicing the local market to a multinational...
-
Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the starting materials you would use, and write thereaction. CHO Cinnamaldehyde
-
What condensation product would you expect to obtain by treatment of the following substances with sodium ethoxide in ethanol? (a) Ethyl Butanoate (b) Cyclopentanone (c) 3, 7-Nonanedione (d)...
-
Solve the following initial value problems: (a) (b) 142 =-111 + 112-2uit 1, 113 =-311 i + 112-2113 + 1, ul (0) = 1 112(0) = 0 113(0) = 0 ul = ul-214 142(0) = 0
-
Nathan Martin has the following data he needs you to include on his Statement of Cashflows Net Income: $32,000 Depreciation: 10,000 Gain on a sale: 8000 Accts Rec decrease: 5,000 Inventory decrease:...
-
Dulux Paints Canada (DPC) is a chain of paint stores in various locations throughout Canada. Fred Antfleck is the sole shareholder of the privately-held company and is looking to expand by either...
-
The Earth's population grows exponentially as N(t)=Ae0.001t. Currently (at t=0) there are 7.9 billion people on Earth. How soon there will be 100 billion people?
-
Your company has the following capital structure that you consider optimal: Ordinary shares Non-redeemable preference shares Debt 50% 15% 35% It is expected that your company's earnings and dividends...
-
Visual Cable TV Company, an accrual basis taxpayer, allows its customers to pay by the year in advance ($350 per year), or two years in advance ($680). In September 2023, the company collected the...
-
Three small, identical \(1.0-\mathrm{kg}\) pucks are attached to identical \(0.50-\mathrm{m}\) strings, tied together at a common center as shown in Figure P11.42. When whirled in circular motion at...
-
After looking at the resources, explain what a spirit image is. Why might looking at a god and/or a human in terms of their spirit be helpful if you want to eliminate some of the divisions between...
-
Use formal charge to identify the better Lewis structure. H H-C= H H=C
-
Solved Problem 7-4 showed that the debromination of (R,R)-2,3-dibromobutane gives cis-but- 2-ene. Draw the same reaction using the (S,S) enantiomer and show that it gives the same diastereomer of the...
-
Predict the elimination products formed by debromination of the following compounds with iodide ion in acetone. Include stereochemistry, and give a correct name for each product. (a)...
-
The following compounds show different rates of debromination. One reacts quite fast, and the other seems not to react at all. Explain this surprising difference in rates. Br KI, acetone Br (CH),C RS...
-
An initially motionless test car is accelerated uniformly to 120 km/h in 7.98 s before striking a simulated deer. The car is in contact with the faux fawn for 0.515 s, after which the car is measured...
-
How do you define operational excellence? What factors are involved in achieving operational excellence? Who (within an organization) is responsible for operational excellence and why is this...
-
f(x) = x In(2x) Find f(x) for the function below....

Study smarter with the SolutionInn App


