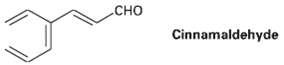
Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the
Question:
Cinnamaldehyde, the aromatic constitution of cinnamon oil, can be synthesized by a mixed aldol condensation. Show the starting materials you would use, and write thereaction.
Transcribed Image Text:
сно CHO Cinnamaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
CHO CH3CHO 1 NaOH EtOH 2 heat ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following compounds can be synthesized by aldol condensations, followed by further reactions. (In each case, work backward from the target molecule to an aldol product, and show what compounds...
-
Epoxides can be synthesized by treating halohydrins with aqueous base. Propose a mecha-nism for reactions (a) and (b), and explain why no epoxide formation is observed in (c). (a) (b) (c) CI NaOH H20...
-
Vicinal halo alcohols (halohydrins) can be synthesized by treating epoxides with HX. (a) Show how you would use this method to synthesize 2-chlorocyclopentanol from cyclopentene. (b) Would you expect...
-
4. Jerry intends to use the money from his loan (and his personal savings if necessary) to make an investment in his friend Elaines business. In return, Elaine has predicted the following returns on...
-
Suppose you were granted a "risky job" of the type studied in this chapter. The job pays $40,000 with probability 1/2, and $160,000 with probability 1/2. What is your certainty equivalent for this...
-
Imagine a container placed in a tub of water, as depicted in the accompanying diagram. (a) If the contents of the container are the system and heat is able to flow through the container walls, what...
-
Describe the six-step IT strategic planning process.
-
Brown Deer Electric sold $3,000,000, 10%, 10-year bonds on January 1, 2012. The bonds were dated January 1 and pay interest July 1 and January 1. Brown Deer Electric uses the straight-line method to...
-
What frameworks can organizations use to ensure ethical decision-making, and how do organizational values and ethics influence stakeholder relationships and corporate reputation ?
-
The comparative financial statements of Automotive Solutions Inc. are as follows. The market price of Automotive Solutions Inc. common stock was $119.70 on December 31, 20Y8. Instructions Determine...
-
How can you account for the fact that 2, 2, 6-trimethylcyclohexanone yields no detectable aldol product even though it has an acidic hydrogen?
-
The bicycle ketone shown below does not undergo aldol self-condensation even though it has two ? hydrogen atoms. Explain.
-
CVI, Inc., had November sales totaling $4,200,000 and incurred the following quality-related costs: Spoiled work-in-process inventory disposal.................................$28,000 Downtime, due to...
-
How can advanced automation tools and artificial intelligence enhance patch management, and what are the potential risks of relying too heavily on automated solutions ?
-
What unique challenges and best practices are associated with patch management in hybrid and multi-cloud environments? Explain
-
In 2023 , John and Peggy Smith purchase and install the following : two exterior doors at a cost of $ 1,000 each , windows and skylights at a total cost of $ 2,200 , and one central air conditioner...
-
How is patch management integrated into your incident response plan, and how do you ensure that patching activities do not impede or complicate forensic investigations during a security incident ?
-
a) Discuss the effect of the change from historical cost to mark-to-market (that is, fair value) accounting on the trade-off between sensitivity and precision of net income as performance measures....
-
A \(0.25-\mathrm{kg}\) ball attached to a string is being spun around on a horizontal surface at a rotational speed of \(2.5 \mathrm{rev} / \mathrm{s}\). What is the ball's kinetic energy if the...
-
For what reason might an exporter use standard international trade documentation (letter of credit, draft, order bill of lading) on an intrafirm export to its parent or sister subsidiary?
-
In N 2 O, nitrogen is the central atom, and the oxygen atom is terminal. In OF 2 , however, oxygen is the central atom. Use formal charges to explain why. Formal charge = number of valence electrons...
-
Give the expected product(s) of E2 elimination for each reaction. (Hint: Use models!) (a) (b) CH, H NaOCH one product HC H NaOCH two products
-
Determine the number of elements of unsaturation in the molecular formula C4H6. Give all nine possible structures having this formula. Remember that A double bond = one element of unsaturation A ring...
-
The preceding example shows meso-1,2-dibromo-1,2-diphenylethane reacting with iodide ion to give trans-stilbene. Show how the other diastereomer of the starting material gives a different...
-
A particle initially has a speed of 0.46c. (Enter your answers in terms of c. Round your answers to at least three decimal places.) (a) At what speed does its momentum increase by 1%? .465 x c (b) At...
-
5) Megan and four of her friends plan to go for a long drive. She buys 12 bottles of frappe and a few bottles of orange juice from a supermarket. The total number of bottles they have are four times...
-
Job Posting Find a job posting for the organization and provide the title of the job posting, including a live hyperlink to the job posting. Apple Support College Program - Appalachian State...

Study smarter with the SolutionInn App


