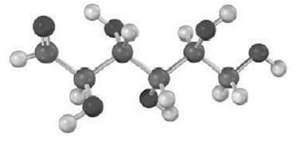
The following model is that of an aldohexose: (a) Draw Fischer projections of the sugar, its enantiomer,
Question:
The following model is that of an aldohexose:
(a) Draw Fischer projections of the sugar, its enantiomer, and a diastereomer.
(b) Is this a D sugar or an L sugar? Explain.
(c) Draw the ? anomer of the sugar in its furanose form.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a OHC HO H A H OH HOH HH OH HOCH H CHO OH OH HOH OH CHOH LMann...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following model is that of an allylic carbocation intermediate formed by Protonation of a conjugated diene with HBr. Show the structure of the diene and the structures of the final...
-
The following model is specified: y1 = 1y2 + 11x1 + 1, y2 = 2y1 + 22x2 + 32x3 + 2. All variables are measured as deviations from their means. The sample of 25 observations produces the following...
-
The following model is specified:y 1 = 1 y 2 + 11 x 1 + 1 ,y 2 = 2 y 1 + 22 x 2 + 32 x 3 + 2 . All variables are measured as deviations from their means. The sample of 25 observations produces...
-
Company OMEGA wants to invest in Bonds and Stocks. The financial manager was looking at different types of Bonds and Stocks. After studying the market, he decided to choose 3 bonds and 3 stocks....
-
You've put a lot of work into your college classes so far-make sure you don't have any glitches as you get ready to claim your certificate or degree. Your task Prepare an interim progress report that...
-
(a) Write the half-reactions for the silver-silver chloride and calomel reference electrodes. (b) Predict the voltage for the following cell. Saturated silver-silver Saturated calomel chloride...
-
Why are costs, expenses, deferrals, and accruals often estimated to a greater extent in interim than in annual financial statements?
-
Nick Waege started his own consulting firm, Waegelein Consulting, on June 1, 2010. The trial balance at June 30 is as follows. In addition to those accounts listed on the trial balance, the chart of...
-
How much work is required to assemble a uniform ball of charge with total (final) charge Q and radius R? Hint: This is the same as the potential energy of the uniform ball of charge. You can compute...
-
On June 1, Depot Company began operations. On September 1, Depot Company has the following accounts and account balances: Cash $17,400, Accounts Receivable $2,000, Supplies $1,900, Accounts Payable...
-
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell whether it is an ? a or ? anomer.
-
Classify each of the following sugars. (For example, glucose is analdohexose.) () CH2 (b) CH2OH (c) - C=0 - C=0 - CH- . - - - CH2 - - - - CH2
-
What are dynamic Web pages?
-
Baker Incorporated had common stock of $150,000 and retained earnings of $850,000. On January 1, 2024,Baker issued 20,000 shares of common stock with a $10 par value and a $40 fair value for all of...
-
What are the efficacy and underlying mechanisms of advanced pharmacological interventions for stress management, and how can clinicians optimize treatment outcomes while minimizing potential side...
-
Provide an alternate perspective on the advantages of using SWOTT over SWOT analysis. Identify an additional aspect of the environmental analysis not covered by SWOTT that would be important to...
-
Few aspects of marketing were of greater concern to healthcare professionals than the onset of advertising. Because of the sensitive nature of healthcare and the often precarious position of the...
-
Determine Net income for tax purposes based on the below criteria and any carry forward amounts? T4 Income $75,000 Business Loss $15,000 Taxable Capital Gains $5,000 Allowable Capital Losses $12,000...
-
The black lines show the emission or absorption lines observed in two spectra of the same element. Which one is an emission spectrum and which is an absorption spectrum? . .
-
Access the Federation of Tax Administrators Internet site at www. taxadmin.org/state-tax-forms and indicate the titles of the following state tax forms and publications: a. Minnesota Form M-100 b....
-
The frictionless piston-and-cylinder system shown here is subjected to 1.013 bar external pressure. The piston mass is 200 kg, it has an area of 0.15 m 2 , and the initial volume of the entrapped...
-
In Figure 6.4 and in the "A Word About...Halogenated Organic Compounds from the Sea," halomon, a polyhalogenated terpene produced by red algae, is shown. a. Assign R or S configuration to the...
-
Draw a Fischer projection formula for the product of this SN2 reaction: CH3 NaSH Br acetone CH2CH
-
Predict the product of the reaction if CH3-S-CH2CH2CH2CH2-Br is heated in a polar organic solvent, such as methanol. Similarly, what would be the product for hexyl bromide in methanol? (In hexyl...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


