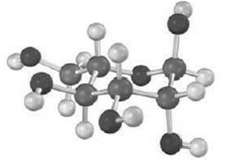
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell
Question:
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell whether it is an ? a or ? anomer.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
The structure represents an o anomer because th...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following structure is drawn in an unconventional manner. (a) Label the N terminus and the C terminus. (b) Label the peptide bonds. (c) Identify and label each amino acid present. (d) Give the...
-
The following structure is an intermediate in a synthesis of prostaglandins F2a and E2 by E. J. Corey (Harvard University). A Horner-Wadsworth-Emmons reaction was used to form the (E)-alkene. Write...
-
The following 1H NMR spectrum is that of an alcohol, C8H10O. Propose a structure. TMS 6. Chemical shift (8) 3 O ppm 10 8. Intensity
-
Patients who undergo chronic hemodialysis often experience severe anxiety. Videotapes of progressive relaxation exercises were shown to one group of patients and neutral videotapes to another group....
-
The use of social networks by employees during work hours remains a controversial topic, with some companies encouraging networking, some at least allowing it, and others prohibiting it. Your task...
-
The space shuttle's expendable booster engines derive their power from solid reactants: 6NH4+ ClO-4 (s) + 10Al(s) 3N2(g) + 9H2O(g) + 5Al2O3(s) + 6HCI(g) (a) Find the oxidation numbers of the...
-
What is a "comprehensive basis of accounting other than GAAP"?
-
On December 31, 2011, Hurly Co. performed environmental consulting services for Cascade Co. Cascade was short of cash, and Hurly Co. agreed to accept a $300,000 zero-interest-bearing note due...
-
1a. Determine this wave's amplitude. 1b. Determine this wave's wavelength. 1c. Determine this wave's frequency. 1d. Determine this wave's speed of propagation. 1e.Determine this wave's direction of...
-
Alexander Smith and his wife Allison are married and file a joint tax return for 2021. The Smiths live at 1234 Buena Vista Drive, Orlando, FL 32830. Alexander is a commuter airline pilot but took 6...
-
Draw Fischer projections of the following molecules, placing the carbonyl group at the top in the usual way. Identify each as a D or Lsugar. (b) la)
-
The following model is that of an aldohexose: (a) Draw Fischer projections of the sugar, its enantiomer, and a diastereomer. (b) Is this a D sugar or an L sugar? Explain. (c) Draw the ? anomer of the...
-
Let T be an n-node binary tree that may be improper. Describe how to represent T by means of a proper binary tree T with O(n) nodes.
-
Evaluate the following expression. 6! 3! 3!
-
(a) Find the gradient of the curve y=2x-7x+4 when x=-2. (b) A is the point (7, 2) and B is the point (-5, 8). (i) Calculate the length of AB.
-
Blue inc. Purchases and assets for $100,000. The asset has a useful life of ten years and no salvage value. In year 6, Blue inc. sold the assets for $60,000. What is the reported gain or loss for...
-
Use power series to evaluate the limit. 7 sin x i. lim x0e2x-1
-
What is the impact of mindfulness-based stress reduction (MBSR) techniques on neuroplasticity, and how do they contribute to long-term stress management ?
-
After the n = 1 to n = 3 absorption shown in the energy-level diagram to the left, which is a possible energy of an emitted photon? A. 1 eV B. 4eV C. 5 eV D. 9 eV
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Find the pH of a 0.350 M aqueous benzoic acid solution. For benzoic acid, K a = 6.5 * 10 -5 . a) 4.64 b) 4.19 c) 2.32 d) 11.68
-
Draw each of the following equations in a way that shows clearly the stereochemistry of the reactants and products. a. (R)-2-bromobutane sodium methoxide (in methanol)2-methoxybutane b....
-
The (+) enantiomer of the inhalation anesthetic desflurane (CF3CHFOCHF2) has the S configuration. Draw a three-dimensional representation of (S)-(+)-desflurane.
-
When treated with sodium iodide, a solution of (R)-2-iodooctane in acetone gradually loses all of its optical activity. Explain.
-
8. A loan is being repaid with a series of payments at the end of each quarter for five years. If the amount of principal in the third payment is $100, find the amount of principal in the last five...
-
If you issue 1000$ of debt and pay it out as dividends, how does it impact your EV?
-
What are APR *and* APY rates of investment X that earns 0.2% per week, compounded weekly? You can invest (that is, 'lend') for one year either at the rate X described above, or, alternatively, at the...

Study smarter with the SolutionInn App


