The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach?
Question:
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach?
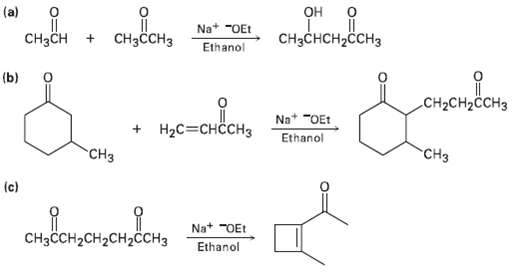
Transcribed Image Text:
о. одо, (a) сые еь он Na* "OEt CHснсH2CсHз CHзсCHз CHзCH Ethanol (b) .oвon CH2CH2CCH3 Na* "OEt + HаС3снсснз Ethanol "СHз "CHз (c) сононсн дсн Na+ "OEt CHзссH-CH2CH2CСH3 Ethanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a Several other products are formed in addition to the one pictured ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
Which of the following reactions are allowed, and which are forbidden, by the conservation laws appropriate to weak interactions? (a) + p + + n, (b) e + p e + + + p, (c) + e + e , (d) ...
-
Which of the following reactions are allowed and which are forbidden by the conservation laws appropriate to weak interactions? (a) + + e + e ; (b) K + + + e + e ; (c) K + 0 + + + ;...
-
Given the following network, with the indicated flow capacities along each branch, determine the maximum flow from source node 1 to destination node 10 and the flow along eachpath: 15 5 10 7 16 12 0...
-
Mercury is a hypothetical store that sells athletic shoes, particularly shoes for runners. Mercury is distinctive in the training of its sales staff. The store has a variety of diagnostic tools,...
-
(a) With reference to Figure 6.18, what is the relationship between the number of nodes in an s orbital and the value of the principal quantum number? (b) Identify the number of nodes; that is,...
-
What is the purpose of a VMO?
-
Afton Co. Purchased $24,000 of 4%, 10-year Davis Country bonds on July 12, 2012, directly from the country at par value. The bonds pay semiannual interest on May 1 and November 1. On December 1,...
-
Partners A, B, and C have capital account balances of $60,000 each. The profit and loss ratio is 5:2:3, respectively. In the process of liquidating the partnership, noncash assets with a carrying...
-
Favorable business conditions may bring about certain seemingly unfavorable ratios, and unfavorable business operations may result in apparently favorable ratios. For example, Sanchez Company...
-
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione? Wieland-Miescher ketone
-
Fill in the missing reagents a?h in the following scheme: CO2Et CH3 CO2Et 2. f 1. c 2. d 1. a 2. b 1. g 1. e 2. h CO2Et
-
The VIF given in Equation 12.67 for a covariate requires computation of R 2 , as obtained in the regression model when the covariate is an output and other covariates are input variables for it....
-
Discuss the security implications of using an RTOS in critical applications. What are some common vulnerabilities, and how does the RTOS architecture address security concerns without compromising...
-
Describe the process of integrating an RTOS into an embedded system. What considerations are there for hardware-specific dependencies, and how does the RTOS interact with various hardware components ?
-
On January 1 , Deacon Co . acquired a 4 0 % interest in Pirate, Inc. for $ 1 , 0 0 0 , 0 0 0 . At the time of purchase, the book value of the net assets of Pirate equaled $ 1 , 8 7 5 , 0 0 0 . The...
-
Explain the differences between task context and interrupt context in an RTOS. How does an RTOS manage interrupt service routines to maintain real-time requirements ?
-
Identify: stimuli that made you recognise a need. want(s) that could fulfil this need. demand(s), i.e., the desire and ability to pay for samsung washing machine to fulfil the need. Internal...
-
A disk of rotational inertia \(I\) about the central axis shown in Figure P11.51 is rotating about this axis with initial rotational velocity \(\omega_{\vartheta, \mathrm{i}}\) on low-friction...
-
Independent random samples of sizes n1 = 30 and n2 = 50 are taken from two normal populations having the means 1 = 78 and 2 = 75 and the variances 21 = 150 and 22 = 200. Use the results of Exercise...
-
Draw the Lewis structure for BrF with an arrow representing the dipole moment. Refer to Figure 10.10 to estimate the percent ionic character of the BrF bond. Percent ionic character 100 75 50 25 0....
-
Predict the major products of acid-catalyzed dehydration of the following alcohols. (a) Pentan-2-ol (b) 1-methylcyclopentanol (c) 2-methylcyclohexanol (d) 2,2-dimethylpropan-1-ol
-
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown. (a) (b) (c) OEt EtOH heat Br OEt CH2OH H SO4 heat...
-
Predict the dehydrohalogenation product(s) that result when the following alkyl halides are heated in alcoholic KOH. When more than one product is formed, predict the major and minor products. (a)...
-
Can you draw a graph to show me how I would depict a change in the Monocentric City Model due to an inflow on the shape of Northwern cities due to the mass migration of African-Americans from the...
-
The firm's production function tell you if a twofold (threefold, etc) increase in all inputs increases output by less than twofold (less than threefold, etc.). Based on this information answer the...
-
After participating in the Application Based Activity (ABA) on the case involving allegations of fraud against the owner of a dating website, I judged the case in favor of the defendant. My decision...

Study smarter with the SolutionInn App


