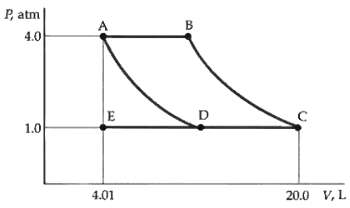
The PV diagram in figure represents 3 mol of an ideal monatomic gas. The gas is initially
Question:
The PV diagram in figure represents 3 mol of an ideal monatomic gas. The gas is initially at point A. The paths AD and BC represent isothermal changes. If the system is brought to point C along the path AEC, find
(a) The initial and final temperatures,
(b) The work done by the gas, and
(c) The heat absorbed by the gas.
Transcribed Image Text:
P, atm 4.0 1.0 20.0 V, L 4.01
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Although not required for this problem we begin by de...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
A 2.0 mol sample of an ideal monatomic gas undergoe the reversible process shown in Figure, the scale of the vertical axis is set by Ts = 400.0K and the scale of the horizontal axis is set by Ss =...
-
The temperature of 2.00 mol of an ideal monatomic gas is raised 15.0 K at constant volume. What are (a) The work W done by the gas, (b) The energy transferred as heat Q, (c) The change Eint in the...
-
In Exercises explain why Rolle's Theorem does not apply to the function even though there exist a and b such that (a) = (b). f(x) H [1,1]
-
Vitex, Inc. manufactures a popular consumer product and it has provided the following data excerpts from its standard cost system: The company's manufacturing overhead cost is applied to production...
-
Discuss when to use active voice and when to use passive voice in sentences. Write a sentence in passive voice then change the sentence to active voice.
-
Bonsai Boards income statement data for the year ended December 31,2010, follow. Assume that the ending inventory was accidentally overstated by $3,300. What are the correct amounts for cost of goods...
-
Millhouse graduated 5 years ago with a degree in business administration and is currently employed as a middle level manager for the same firecracker company his dad already worked for. His current...
-
Identify three to five specific budgeting objectives for a mental health center (these should be specific, measurable, achievable, realistic, and have a time element).
-
1. What is monogenism? Why is it important for our understanding of the human race? 2. According to our lecture, there are 2 ways to order life (put it in a hierarchy). What are these 2 ways, and...
-
What is the number of moles n of the gas in Problem79? P, atm A 4.0 1.0 4.01 20.0 V, L
-
Repeat Problem 81 with the gas following path ABC. P, atm 4.0 1.0 20.0 V, L 4.01
-
Prepare Statement of Cash Flows, Indirect, IFRS. Use the information from E22-1 assuming Hockey Apparel Providers, Inc. is an IFRS reporter. Compute cash flows from operating activities for Hockey...
-
Run the following code to create a dataframe object dr and assign a dataset which contains the sales achievement of the salemen in different stores. Then write code to complete the tasks below. Store...
-
Click the + icon to the left of the Flags. Look at the source and destination ports and the flags that are set. 9. *Wi-Fi File Edit View Go Capture Analyze Statistics Telephony Wireless Jools Help...
-
No. top 1. Time 2. 1105 01:50:04.676815 1117 01:50:05.727813 1118 01:50:05.762296 1119 01:50:05.762546 1129 01:50:05.763502 1121 01:50:05.80G063 3. [TCP Segment Len: 0] Sequence number: 0 [Next...
-
The following figure and table are the process address space and machine state when CPU is about to fetch and execute instruction at Ox80483dd push %ebp. Please describe what changes will be made to...
-
1:24 Back Hwrk07.pdf Q Homework 07 COMSC-122 Homework-07A You will find two files in your Homework07 dropbox: GirlNames.txt - which is a file containing a list of the 200 most popular names given to...
-
A governor is said to be sensitive, when: (a) The ratio of the difference of maximum and minimum equilibrium speeds to mean speed is maximum (b) Governor readily responds to a small variation of...
-
Burberrys competitive advantage is through its differentiation strategy. What risk should Burberry remain aware of?
-
How does the power of a dry contact lens compare with its power when resting on the tear layer of the eye? Explain.
-
In Example 21.3, calculate the net force on charge q1.
-
In Example 21.4, what is the net force (magnitude and direction) on charge q1 exerted by the other two charges?
-
Three point charges are arranged along the x-axis. Charge q1 = +3.00C is at the origin, and charge q2 = -5.00C is at x = 0.200 m. Charge q, = - 8.00 C. Where is q3 located if the net force on q1 is...
-
Calculate the missing information in the STR Report. Round your answers to 1 decimal place. My Prop. Occupancy Comp. Set ADR REVPAR Index My. Prop. Comp. Set Index My Prop. Comp. Set. Current Month...
-
Problem 1 Emma Li graduated from university five years ago with an undergraduate degree in finance. Emma currently works as an investment analyst at a private equity firm. Her annual salary at the...
-
5. The shaft shown in the figure below is proposed for the application as a gear reduction unit. The material is AISI 1018 cold- drawn steel. The gears seat against the shoulders, and have hubs with...

Study smarter with the SolutionInn App


