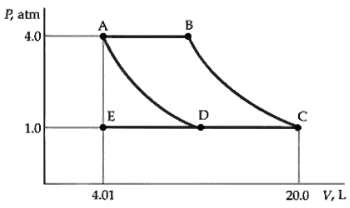
What is the number of moles n of the gas in Problem79? P, atm A 4.0 1.0
Question:
What is the number of moles n of the gas in Problem79?
Transcribed Image Text:
P, atm A 4.0 1.0 4.01 20.0 V, L
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Equation 1916 W nRT ln V ...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
What will happen to the number of moles of SO3 in equilibrium with SO2 and O2 in the reaction 2SO3(g) 2SO2(g) + O2(g) in each of the following cases? a. Oxygen gas is added. b. The pressure is...
-
What is the number of times interest charges are earned if the business has net income before taxes of $600,000 and a $1,500,000 face value bond payable with a coupon rate of 10%?
-
In Exercises sketch the graph of an arbitrary function that satisfies the given condition but does not satisfy the conditions of the Mean Value Theorem on the interval [-5, 5]. is continuous on...
-
SkyChefs, Inc., prepares in-flight meals for a number of major airlines. One of the company's products is grilled salmon in dill sauce with baby new potatoes and spring vegetables. During the most...
-
What is the difference between an NAP and an NSP?
-
The Oasis Hotel is planning its cash payments for operations for the fourth quarter (October-December), 2003. The Accrued Expenses Payable balance on October 1 is \($136,000.\) The budgeted expenses...
-
The Ferre Publishing Company has three service departments and two operating departments. Selected data from a recent period on the five departments follow: The company allocates service department...
-
On August 1, 2027, Pronghorn Corporation issued $494,400,8%, 10-year bonds at face value. Interest is payable annually on August 1. Pronghorn's year-end is December 31. Prepare a tabular summary to...
-
Snookers Restaurant is open from 8:00 am to 10:00 pm daily. Besides the hours that they are open for business, workers are needed an hour before opening and an hour after closing for setup and...
-
In the process of compressing n moles of an ideal diatomic gas to one-fifth of its initial volume, 180 kJ of work is done on the gas. If this is accomplished isothermally at room temperature (293 K),...
-
The PV diagram in figure represents 3 mol of an ideal monatomic gas. The gas is initially at point A. The paths AD and BC represent isothermal changes. If the system is brought to point C along the...
-
Hydrogen iodide is a reactive compound that is sometimes used in the manufacture of methamphetamine. It can be produced by the direct reaction of the elements. Calculate the standard Gibbs free...
-
At the end of the current year, Accounts Receivable has a balance of $870,000, Allowance for Doubtful Accounts has a debit balance of $8,000, and sales for the year total $3,920,000. Bad debt expense...
-
what ways can adaptive linguistic frameworks be leveraged to navigate the complexities of cross-cultural communication and facilitate meaningful global collaborations?
-
The following information is available for Water Inc. Date January 1 (beginning inventory) 440 Purchases: January 10 January 15 January 30 Units Unit Cost $50.00 330 52.00 660 52.50 440 55.00 The...
-
1. A pilot drops a bottle out of an airplane flying 500 m into the ocean. The bottle lands 400 m horizontally from the initial dropping point. How fast was the plane flying when the bottle was...
-
what ways can strategic crisis communication and reputation management minimize the negative impact of unexpected business disruptions or public relations challenges?
-
Based on Exhibit 1, if Yetas management implemented Proposal #3 at the current share price, earnings per share would: A. decrease. B. remain unchanged. C. increase. John Ladan is an analyst in the...
-
Briefly discuss the implications of the financial statement presentation project for the reporting of stockholders equity.
-
What power of spectacle lens is needed to allow a farsighted person, whose near point is 1.00 m, to see an object clearly that is 25.0 cm away? Assume the spectacle (corrective) lens is held 1.50 cm...
-
A negative charge -0.550C exerts an upward 0.200-N force on an unknown charge 0.300 m directly below it. (a) What is the unknown charge (magnitude and sign)? (b) What are the magnitude and direction...
-
Three point charges are arranged on a line. Charge q3 = + 5.00 nC and is at the origin. Charge q2 = - 3.00 nC and is at x = +4.00cm. Charge q1 is at x = +2.00cm. What is q1 (magnitude and sign) if...
-
In Example 21.4, suppose the point charge on the y-axis at y = -0.30 m has negative charge -2.0, C, and the other charges remain the same. Find the magnitude and direction of the net force on Q. How...
-
Loving Gardens (LG) has $8 million in assets, $600,000 EBIT, and a marginal tax rate equal to 40 percent. If LG's debt ratio (D/TA) is 50 percent, interest on its debt is 10 percent, whereas if the...
-
You are the vice-president of finance for Exploratory Resources, headquartered in Calgary. In January 2012, your firm's American subsidiary obtained a six-month loan of $2.5 million (U.S.) from a...
-
1. Calculate the monthly payment for a home loan of $800,000 financed at 8.0% over 30 years. na ab appula inter Present Value (PV) Payment (PMT) Future Value Annual Rate (FV) (I) Compounding Periods...

Study smarter with the SolutionInn App


