There are four resonance structures for anthracene, one of which is shown. Draw the otherthree. Anthracene
Question:
There are four resonance structures for anthracene, one of which is shown. Draw the otherthree.
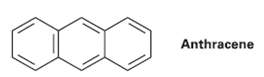
Transcribed Image Text:
Anthracene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
For your analysis, choose 10 countries, one of which is the United States. Create a table showing whether each country applies a worldwide or territorial approach to international income taxation....
-
For your analysis, choose 10 countries, one of which is the United States. Create a table showing whether each country applies a worldwide or territorial approach to international income taxation....
-
There are two contributing resonance structures for an anion called acetaldehyde enolate, whose condensed molecular formula is CH2CHO-. Draw the two resonance contributors and the resonance hybrid,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Recruiting people for jobs that require international assignments is increasingly important for many organizations. Where might an organization go to recruit people interested in such assignments?
-
To estimate the proportion of troops who support the U.S. militarys Dont Ask, Dont Tell policy of discharging people who are openly homosexual, a political researcher interviewed a random sample of...
-
Indicate whether each of the following statements constitutes a potential advantage (A), disadvantage (D), or neither (N) of using transfer prices for support department costs. a. Requires additional...
-
Your uncle offers you a choice of $105,000 in 10 years or $47,000 today. If money is discounted at 9 percent, which should you choose?
-
Assume that the cooperative has $1,645,371 of Net Income to allocate to patrons ($1,785,321 Net Income - $139,950 actual Unallocated Reserve contribution). Also assume that 30%, or $493,611, of this...
-
The second picture is the needed data from P2 that P3 says is needed P3. Consider a possible shortage of ingredients in the following week. What would the optimized production process look like if...
-
Look at the three resonance structures of naphthalene shown in Section 15.7, and account for the fact that not all carboncarbon bonds have the same length. The C1C2 bond is 136 pm long, whereas the...
-
There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour. Phenanthrene
-
Describe the up-front television buying for prime time.
-
Kyle Gamble is a sales manager. Kyle is 48 years old and is married to Kara who is 45 years old and blind. She has 2021 net income of $9,000, all of which is interest on investments she inherited...
-
Question 17: Task 17: Role play See detailed handout on the exemplar report and its background Read and discuss the report in small groups Discussion in full group: Audience Structure Data and...
-
HomeLife Life Insurance Company has two service departments (actuarial and premium rating) and two production departments (advertising and sales). The distribution of each service department's...
-
1. 2. 3. Suppose that the long-run world demand and supply elasticities of crude oil are -0.906 and 0.515, respectively. The current long-run equilibrium price is $30 per barrel and the equilibrium...
-
ANSWER THE FOLLOWING QUESTION AND PROVIDE SOLUTIONS : Problem 9.1 COMPREHENSIVE Valkyrie reported the following income during the year. Service Fees Interest Income from bank deposits Royalties from...
-
Discuss how both unions and firms can be better off if they move off the demand curve. Derive the contract curve.
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
The equilibrium constant for the reaction A(g) B(g) is 10. A reaction mixture initially contains [A] = 1.1 M and [B] = 0.0 M. Which statement is true at equilibrium? (a) The reaction mixture...
-
Draw the complete structures of the following peptides: (a) Thr-Phe-Met (b) Serylarginylglycylphenylalanine (c) IMQDK (d) ELVIS
-
Draw the structure of the phenylthiohydantoin derivatives of (a) Alanine (b) Tryptophan (c) Lysine (d) Proline
-
Show the third and fourth steps in the sequencing of oxytocin.
-
(a) A friend says "Insurance policies are a rip-off; they always have provisions that limit how much you can be compensated." You will enlighten your friend by saying . . . (b) Nestle Ghana has the...
-
For tax purposes, sole proprietorships: are not taxed, and the owner is not taxed on their distribution. are pass-through entities. pay taxes on the income of the business, and the owner is taxed on...
-
Complete Marked out of 1.00 Flag question Question text What pre-balance checks should be made to wheels prior to carrying out a wheel balance? wheels should be carefully inspected for dents, bent or...

Study smarter with the SolutionInn App


