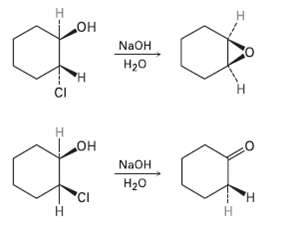
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the
Question:
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the same conditions yields Cyclohexanone. Propose mechanisms for both reactions, and explain why the different results areobtained.
Transcribed Image Text:
н но° N2OH H20 н но NAOH Н2о 'CI Н Н Н т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
H OH trans2Chloro cyclohexanol ring flip CI H OH cr H H 12Epoxyeyelohexane H cis2Chl...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose mechanisms for the reactions shown in Problems 22-62 parts (a) and (b) and 22-63 parts (a) and (b). In problem 22-62 (a) (b) In problem 22-63 (a) (b) CH3 TOH CH CH2-C-H CH CH OCH3 CH3OH CH3...
-
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
-
Propose reasonable fragmentation mechanisms that explain why The EI mass spectrum of benzoic acid shows major peaks at m/z = 105 and m/z = 77.
-
The costs of achieving emission reductions in the future will depend greatly on the types of policies used to reduce emissions today. Explain.
-
Which of Porter's Five Forces did Apple address through the introduction of the iPhone and customer developed iPhone applications?
-
The current order quantity for Electric Powerbars is 100 bars. The order cost is $10 per order, the holding cost is $0.25 per box per year, and the annual demand is 2,000 bars per year. a. Calculate...
-
The following questions are included in Figure 10-3: 1. Are hirings, pay rates, payroll deductions, and terminations a:t thorized in writing by the personnel department? 2. Are payroll deduction...
-
Elliott Company produces large quantities of a standardized product. The following information is avail-able for its production activities for March. Additional information about units and costs of...
-
a) Figure 4 shows a PLD with 3 inputs and 2 outputs before it is programmed. Redraw the PLD after being programmed to produce these Boolean expressions: F1 = A'B'C'+ABC and F2 = A'BC+ABC. (5 marks)...
-
At the breakeven point, a company's sales revenue equals its expenses and there is zero profit. Breakeven analysis can be expanded to encompass the analysis of costs, volume and profit, so that a...
-
Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason...
-
Ethers undergo an acid-catalyzed cleavage reaction when treated with the Lewis acid BBr3 at room temperature. Propose a mechanism for thereaction. + CHBr CH 1. r 2. H20
-
When will the world run out of oil? One way to judge is to determine the oil reserves of the countries around the world. The next table displays the known oil reserves of the top 15 countries....
-
The Cutting Department of Lasso Company has the following production and cost data for August. Production Costs 1. Started and completed 9,600 units. Beginning work in process $0 2. Started 2,300...
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
5 5 Color the World Co., with a December 31 year end date, started to sell high definition televisions for $2,000 each on Jan 1, 2018. Each television includes one year warranty covering parts and...
-
Shea Jonas, a fellow student, is unclear about the basic steps in the recording process. Identify and briefly explain the steps in the order in which they occur. Indicate basic and debit-credit...
-
Classify each of the following activities as proper or prohibited under the various consumer statutes you have studied. a. Calling a hospital room to talk to a debtor who is a patient there. b....
-
Hydrogen has an auto-ignition temperature of 853 K; that is, hydrogen will ignite spontaneously at that temperature if exposed to oxygen. Hydrogen is to be adiabatically and reversibly compressed...
-
Write bond-line structural formulas for (a) Two primary alcohols, (b) A secondary alcohol, and (c) A tertiary alcohol-all having the molecular formula C4H10O.
-
One way of naming alcohols is to name the alkyl group that is attached to the -OH and add the word alcohol. Write bond-line formulas for (a) Propyl alcohol and (b) Isopropyl alcohol.
-
One way of naming ethers is to name the two alkyl groups attached to the oxygen atom in alphabetical order and add the word ether. If the two alkyl groups are the same, we use the prefix di-, for...
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...
-
An 84 year old female arrives to the ER today with her son with a complaint of new onset of confusion. What are your differential diagnoses for a patient with new altered mental status? Go through...

Study smarter with the SolutionInn App


