Which of these compounds will have more of the conformation with the substituent on the cyclohexane ring
Question:
Which of these compounds will have more of the conformation with the substituent on the cyclohexane ring axial present at equilibrium?
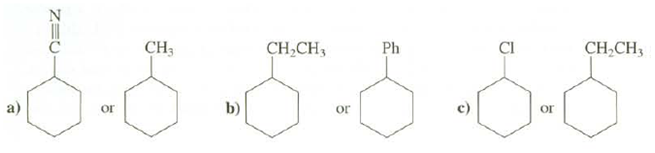
Transcribed Image Text:
CH,CH3 CI CH3 Ph CH,CH, b) or or or
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
Substituents larger than H prefer to be equatorial on a cyclohexane ring Larger substituents have a ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist? a) CHCHCH=CHCH CH3 c) CHC=CHCH b) CH3CHCH=CH CI d) CHC=CHCHCH3
-
Which of these compounds can form an intra molecular (within the same molecule) hydrogen bond between the hydrogen of the carboxylic acid and the oxygen of the ether group? Explain. H, H, H -C-c-c- |
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Find f. f(x) = f'(x) = 12x + X x > 0, f(1) = -4
-
Kevin Ltd is developing departmental overhead rates based on machine hours for its Moulding Department and direct labour hours for its Assembly Department. The Moulding Department has 20 machines,...
-
Diane's Dairy Sales & Delivery finished its first year of operations on December 31, 2014. After adjusting journal entries, the company presented the following adjusted trial balance. Using this...
-
Using information from Exercise 22, create an alphabetized listing by Interface ID that describes the interactions that occur across each interface by identifier. Exercise 22 Create an N2 Diagram for...
-
The following audit report was drafted by a junior staff accountant of Lipske & Griffin, CPAs, at the completion of the audit of Douglas Companys ICFR. The report was submitted to the engagement...
-
Minden Company is a wholesale distributor of premium European chocolates. The company s balance sheet as of April 3 0 is given below: Minden Company Balance Sheet April 3 0 Assets Cash $ 1 1 , 4 0 0...
-
Code in C only. Chef is very fond of horses. He enjoys watching them race. As expected, he has a stable full of horses. He, along with his friends, goes to his stable during the weekends to watch a...
-
Draw the two chair conformations for ethyl cyclohexane which is more stable.
-
Bromine is larger than chlorine, yet the two atoms have identical axial destabilization energies. Explain.
-
The ionization of the hydrogen atom can be calculated from Bohrs equation for the electron energy. where NARhc = 1312 kJ/mol and Z is the atomic number. Let us use this approach to calculate a...
-
If f(x) is defined for all x and f (x)= x(x-4)2, then the critical values for f(x) are: a- 0,-4 c-0.4 d-4,-4 For which values of x is the rate of change of fix)-4x -0.5x +1000 increasing: 0
-
Calculate a one-year holding period return (HPR) for the following two investment alternatives: E. Which investment would you prefer, assuming they are of equal risk? Explain. The HPR for investment...
-
1. A mechanic lifts a 1740 kg car using a hydraulic lift. The lift uses two pistons with diameters in ratio of d/d = 5.30, with the car lifted using the large piston to a height of 1.75 m. (a) How...
-
Each group is required to download and use the latest financial annual report (FYE 2022) of the selected company and refer to the "Group" / "Consolidated" financial data to answer the following...
-
Date CAT MOT 01-Dec-05 52.09 21.51 01-Nov-05 52.1 22.9 03-Oct-05 47.42 21.06 01-Sep-05 52.74 20.94 01-Aug-05 49.81 20.76 01-Jul-05 48.39 20.1 01-Jun-05 42.58 17.33 02-May-05 42.04 16.45 01-Apr-05...
-
How is scrap accounted for?
-
You deposit $10,000 in a savings account that earns 7.5% simple interest per year. What is the minimum number of years you must wait to double your balance? Suppose instead that you deposit the...
-
Find the area enclosed by the line y = x 1 and the parabola y 2 = 2x + 6.
-
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent? (b) (a)
-
Write the mechanism of the reaction between p-hydroxyaniline and acetic anhydride to prepare acetaminophen.
-
What product would you expect from reaction of 1 equivalent of methanol with a cyclic anhydride, such as phthalic anhydride (1, 2-benzenedicarboxylic anhydride)? What is the fate of the second ?half?...
-
1. Answer the following questions: Suppose you are given the following tabular dataset: Code Title Pages Study Level Price Availability 525745 Introduction to Java 256 Easy 57.20 Yes 525746 Advanced...
-
What is wrong with this code & why? How do I fix it? public class JavaApplication1 { private int num; public static void main(String[] args) { num = 14; num = num ++ % 3 + num; System.out.println(...
-
A classifier was built to distinguish between 3 types of news articles: S, H, and P. After training, it was run over 15 testing news articles and here are the results: Article # Actual type Predicted...

Study smarter with the SolutionInn App


