Vanillin (the natural vanilla flavoring) occurs in nature as a -glycoside of glucose. Suggest a structure for
Question:
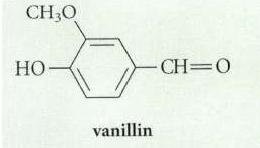
Transcribed Image Text:
CH,O vanillin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Because naturally occurring glycosides generally ha...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Vanillin is the active component of natural vanilla flavoring. OCH vanillin
-
Pectin, which occurs in plant cell walls, exists in nature as a polymer of D-galacturonic acid methylated at carbon 6 of the monomer. Draw a Haworth projection for a repeating disaccharide unit of...
-
Vanillin, the dominant flavoring in vanilla, contains C, H, and O. When 1.05 g of this substance is completely combusted, 2.43 g of CO2 and 0.50 g of H2O are produced. What is the empirical formula...
-
The adjusted trial balance for Ray Corporation at July 31, 2017, the corporation's fiscal year end, contained the following: Of the lease liability amount, $16,250 is due within the next year. Total...
-
Esther (All) God is not mentioned in this book. How does Gods providence show up in this book?
-
Athletes who win Olympic gold medals also receive a cash award of $ 25,000. In 2012, President Obama supported legislation proposed by Senator Marco Rubio to exempt these winnings from federal...
-
Using any of the datasets that come with this text that include at least two quantitative variables and at least one categorical variable (or any other dataset that you find interesting and that...
-
Cochrane, Inc., is considering a new three-year expansion project that requires an initial fixed asset investment of $2.7 million. The fixed asset will be depreciated straight-line to zero over its...
-
3. Solve system of linear equations using either Gaussian elimination with back-substitution. Show your work (explain what equations you are multiplying and/or adding when apply). 4x+5y-5z = -23 -2y...
-
8. Jack Whitcombe and Sons is a consultant engineering firm. The accounts for the firm are as follows. Bank Car Expense HST Recoverable HST Payable J. Whitcombe, Capital J. Whitcombe, Drawings Fees...
-
Into what other aldose and 2-ketose would each of the following aldoses be transformed on treatment with base? Give the structure and name of the alssose, and the structure of the 2-ketose. D-allose
-
Draw structures for: Is opropy 1-D-galactopyranoside
-
Assume that we want to use the sample data from Exercise 1 to test the claim that the sample is from a population with a standard deviation less than 1.8 min; we will use a 0.05 significance level to...
-
List at least 3 controls / procedures that you could adopt in a business for the handling of electronic payments .
-
How does the strategic use of allusions contribute to the thematic coherence and aesthetic unity of literary works, weaving together disparate strands of cultural heritage and intellectual history to...
-
who report the capital gain and dividends in shares under age?
-
Additional Credits and Payments, what new line was added to report the Credit for Previously Owned Clean Vehicles?
-
In what ways do allusions serve as rhetorical devices, imbuing texts with resonance and depth by evoking shared cultural knowledge, values, and experiences, while also establishing connections...
-
What is gain sharing?
-
A copper sphere of 10-mm diameter, initially at a prescribed elevated temperature T;, is quenched in a saturated (1 atm) water bath. Using the lumped capacitance method, estimate the time for the...
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
Image transcription text Introduction: The Smart Parking Management System (SPMS) project aimed to develop an efficient, automated solution for managing parking spaces in urban areas using Arduino...
-
A. Can we use human capital theory to explain "Age-Discrimination"? If so, how? B. Why do some firms prefer to hire younger workers, especially for entry level positions? C. Why do some firms...
-
Image transcription text Question 6 Not yet answered Marked out of 1.00 '1" Flag question During a mountain?biking tripr Bill and Barry are discussing the merits of having the springs and shocks used...

Study smarter with the SolutionInn App


