What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b)
Question:
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation?
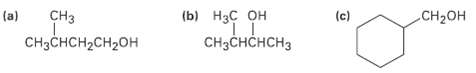
Transcribed Image Text:
(a) CH3 (b) НаС он CHзснснсHз (c) .CH-он CHзCHCH2CH2OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
Strategy As described in Worked Example 72 the strategy in this sort ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What variables might be used to segment these industrial markets? (a) Industrial sweepers, (b) Photocopiers, (c) Computerized production control systems, (d) Car rental agencies.
-
What approaches might be used to recognize risk in capital budgeting?
-
What arguments might be used to persuade shareholders in an acquisition target to reject offers at any price? Are they plausible?
-
Draw isometric projection of the following figure 10 40 50 $30 Pg. 3 : 20
-
Consider the following Excel regression of perceived sound quality as a function of price for 27 stereo speakers. (a) Is the coefficient of Price significantly different from zero at = .05? (b) What...
-
A bacterial population following b(t) = (1.0 + 2.0t)3. For each equation for population size, find the following. a. The average rate of change between times 0 and 1.0. b. The average rate of change...
-
Justify what general factors you consider should be taken into account when recruiting salespeople for the positions described in the exercise. In particular, suggest how the performance of such...
-
These financial statement items are for Major Company at year-end, July 31, 2012. Instructions (a) Prepare an income statement and an owner?s equity statement for the year. The owner did not make any...
-
Use the following information to create a budget for the Constantine family. The Constantine family recently had a budget deficit, something that they want to avoid because they understand that it...
-
Tuff Wheels was getting ready to start its development project for a new product to be added to its small motorized vehicle line for children. The new product is called the Kiddy Dozer. It will look...
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structures of both, and explain the result.
-
A Web ad can be designed from four different colors, three font types, five font sizes, three images, and five text phrases. A specific design is randomly generated by the Web server when you visit...
-
Based on positive results in DT&E and following a successful Operational Test Readiness Review, the SRAW entered IOT&E. The IOT&E plan called for three phases with the first phase (20 shots) being...
-
This is a research to investigate the FAA maintainability requirements for aviation systems. Begin by considering the following statement from the United States General Accounting Office Testimony,...
-
Jenny Corporation recorded warranty accruals as at December 3 1 , 2 0 1 9 , in the amount of $ 1 5 0 , 0 0 0 . This reversing difference will cause deductible amounts of $ 5 5, 0 0 0 in 2 0 2 0 , $...
-
Acme Company uses process costing. Here are data regarding the first processing department for the current month: Work in process inventory, beginning Units Direct material costs 2,700 units $14,800...
-
Moody Corporation uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company made the following estimates:...
-
Describe the three major kinds of plate boundaries.
-
If the cylinder described in Problem 21.3 were initially heated to 500F, how long would it take for the center of the cylinder to cool to 240F if it were constructed of a. Copper? b. Brass? c. Nickel?
-
What is the dipoledipole force? How can you predict the presence of dipoledipole forces in a compound?
-
Rank the compounds within each of the sets shown in Fig. P7 .62 according to their heats of formation, loweet first. Explain. Fig. P7 .62 (a) (b) , H CH3 CH3 H CH
-
(a) What two diastereomeric products could be formed in the hydroboration-oxidation of the following alkene? (b) Considering the effect of the methyl group on the approach of the borane-THF reagent...
-
The ÎG° for the equilibrium in Fig. P7.67ais 4.73 kJ mol-1 (t.13 kcal mol-1. (The equilibrium favors conformation A.) (a) Which behaves as if it is larger, methyl or phenyl (Ph)? iWhy is...
-
Suppose you bought a new home for $210,000 using a 30-year mortgage with monthly payments of $1,218.841. The annual interest rate of the mortgage is 5.7%. After the first 3 years (36 monthly...
-
Rather than use the hypothetical information above, determine from available news sources if the Fed is increasing, holding steady, or decreasing interest rates. Similarly, is the Federal Government...
-
For a two-sided matching problem, we say that matching is Pareto efficient, if there is no matching ', such that and '(x) R (x) for all x WUF, '(x) Px (x) for some x WUF. Show that stability implies...

Study smarter with the SolutionInn App


