What bonds angles do you expect for each of the following, and what kind of hydribization do
Question:
What bonds angles do you expect for each of the following, and what kind of hydribization do you expect for the central atom ineach?
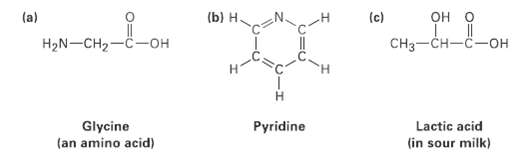
Transcribed Image Text:
(a) (b) H. OH O (c) H2N-CH2-C-OH CHз—CH—с—он Lactic acid (in sour milk) Glycine (an amino acid) Pyridine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a H120 OH NH Gl...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What kind of hydribization do you expect for each carbon atom in the followingmolecules? CH- (b) H. C-H (a) 1 "CH-CH CH2 C=C H2N H. Procaine Vitamin C (ascorbic acid)
-
What kind of hybridization do you expect for each carbon atom in the followingmolecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, CH CH3C=CH2 (c) 1-Butene 3 yne, H2C=CH-C=CH (d) Acetic acid, CHC
-
What new formula results for each of the following formulas if you copy it from cell C10 to cell E12? a. =A1+A2 b. =$A$1+A2 c. =$A1+A2 d. =A$1+A2
-
Figure 20.3 describes the Medicare Part D prescription drug benefi t. Look at the Web page www.partd-medicare.com/ in your area and determine the marginal coinsurance rates applicable in each...
-
The taxpayer's ending inventory is valued as follows: Item Cost Market Rakes...................$8,100......................$7,900 Shovels................$3,800......................$4,300...
-
Mutations can also be caused chemically, and nitrous acid is one of the most potent chemical mutagens. One explanation that has been suggested for the mutagenic effect of nitrous acid is the...
-
A normal shock propagates at \(2000 \mathrm{ft} / \mathrm{s}\) into the still air in a tube. The temperature and pressure of the air are \(80^{\circ} \mathrm{F}\) and 14.7 psia before "hit" by the...
-
On April 1, Wonder Travel Agency Inc. was established. These transactions were completed during the month. 1. Stockholders invested $30,000 cash in the company in exchange for common stock. 2. Paid...
-
Give some example of aggressive behaviour?
-
Rainbow Life Insurance Company, based in Toronto, uses a job evaluation system of their own design. The system is based on five job characteristics: Skill, Mental Effort, Physical Effort,...
-
What is the shape of benzene, and what hydribization do you expect for eachcarbon? C=c H-C - Benzene C-C I.
-
Convert the following structures into skeletal drawings: (a) (b) H. H. H. Indole 1,3-Pentadiene (d) (c) H. -CI C-CI H. 1,2-Dichlorocyclopentane Benzoquinone U
-
Why does A-1 Student Auto Insurance Company need to hold more liquid assets than Senior Life Insurance Company? How do depository institutions manage liquidity risk?
-
What would be the total price ABC should pay for the 350 units for an 80% learning curve for labor, for a 90% learning curve and for a 95% learning curve for labor? What % difference is this between...
-
The board is expecting a regular growth each year. Our expert says that, although not certain, growth between -2% and 5% per year is very reasonable. So we have to make some preparations for this...
-
In a hierarchical organization, each person (except the ranking officer) reports to a unique superior officer. The reporting hierarchy can be described by a tree T, rooted at the ranking officer, in...
-
Consider the following sales data measured in millions of dollars. Date Time Sales (millions of dollars) MA CMA Seasonal Factors Seasonal Index 2015Q1 1 14 2015Q2 2 16 2015Q3 3 18 2015Q4 4 30 2016Q1...
-
Does China benefit from a weak currency?Does Us benefit from a strong currency? Why? Please explain
-
Based on Exhibit 1 and relative to the benchmark, the manager of Fund 1 most likely used a: A. growth tilt. B. greater tilt toward small cap. C. momentum-based investing approach. Stephanie Tolmach...
-
Classify each of the following as direct costs or indirect costs of operating the Pediatrics ward for children at the Cleveland Clinic: a. Wi-Fi covering the entire hospital campus b. Net cost of...
-
It has been suggested that a simple cubic equation of state can also be used to describe a solid. However, since in a solid certain molecular contacts are preferred, compared with a fluid (vapor or...
-
Determine the degree of un-saturation for these compounds.
-
Draw a 3s atomic orbital and compare it to a 2s orbital.
-
Show an atomic orbital energy level diagram for these atoms: (a) Si (b) Al (c) Cl
-
On a particular day, there were 300 stocks that advanced on the NYSE and 800 that declined. The volume in advancing issues was 1000 and the volume in declining issues was 3000. What is the trin ratio?
-
A coupon bond that pays interest of $30 annually has a par value of $1000, matures in 6 years, and is selling today at $850. What is the yield to maturity on this bond?
-
A callable bond pays annual interest of $40, has a par value of $1000, matures in 4 years but is callable in 3 years at a price of $1100, and has a value today of $1020. What is the yield to call on...

Study smarter with the SolutionInn App


