What two constitutional isomers could be formed in the following Diels-Alder reaction? CO,CHs H,C CH2
Question:
What two constitutional isomers could be formed in the following Diels-Alder reaction?
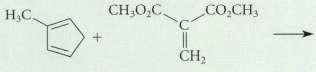
Transcribed Image Text:
CO,CHs H,C CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
t wo possible orientations of ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
Two constitutional isomers of molecular formula C8H12O are formed in the following reaction. Ignoring stereochemistry suggest reasonable structures for these Diels-Alder adducts. H.C
-
Compare the following two constitutional isomers. The 13 C NMR spectrum of the first compound exhibits five signals, while the second compound exhibits six signals. Explain. .
-
You borrowed $325000 using a 30- year fixed rate mortgage with a 5.25% interest rate: A) What is your schedule monthly payment? B) What is the amount of interest and principal paid with the first...
-
The following data (in thousands of dollars) have been taken from the accounting records of Karmana Corporation for the just completed year. (This company puts all manufacturing overhead into work in...
-
Prichards sales in the year ended 31 December 2011 were 390,000. The sales produced a gross profit ratio of 30%. (a) If the cost of inventory on 31 December 2010 was 78,000 and that of 31 December...
-
Why did Europeans colonize regions in the 19th century and how did they justify taking colonial possessions?
-
Seigel, a seventy-three-year-old man, was injured at one of Giant Foods retail food stores when a bottle of Coca-Cola exploded as he was placing a six-pack of Coke into his shopping cart. The...
-
Locate any points of inflection. Enter only the x-value of the solution! Enter if there is no inflection point. -f(x)=-3x-27x2+2x+5
-
Which histograms are skewed to the left? Refer to histograms A through H in Figure 2.12. 2.0 -50 0.0 1.0 3 150 5 15 25 D -1 -3 8 14 -7 -4 -1 G Figure 2.12 Eight histograms LO 09 007 01 08 01 250 09...
-
What product would be expected from the Diels-Alder reaction of 1, 3-butadiene and ethylene?
-
Complete the following Diesls-Alder reaction NC CN NC CN
-
Show that the dc component is the dominant spectral line for high PRF waveforms.
-
Lorna, Melva and Donna are three sisters and shareholders in Growing Green Limited, a private company limited by shares. The company was incorporated in May 2014, with 1000 shares, and specializes in...
-
QUESTION 3: Matters Affecting Enforceability of Contract (suggested word account 800 word maximum) 35 marks Zig and Yul have been negotiating the sale and purchase of Zig's accounting business. As...
-
This essay is on the film KING CORN. Your essay must answer the following questions in essay form (NOT short/long answer) , using APA formatting and citations. 3-5 pages not counting cover and...
-
Which cultural dimension causes the most conflict in your friendships and/or family relationships? Explain how the cultural dimension works as though you were explaining it to someone who has never...
-
sExplore the relationship between deadlock and resource management policies. How can proper resource management help in reducing the likelihood of deadlock in large-scale systems?
-
a. What is the relationship between the value of an unlevered firm and the value of a levered firm once we consider the effect of corporate taxes? b. If we only consider the effect of taxes, what is...
-
You are standing at x = 9.0 km and your assistant is standing at x = 3.0 km. Lightning bolt 1 strikes at x = 0 km and lightning bolt 2 strikes at x = 12.0 km. You see the flash from bolt 2 at t = 10...
-
Doreen Dimwhistle has proposed the following variations on the Chichibabin reaction: She is shocked to find that neither of these reactions works as planned and has come to you for an explanation....
-
At 368C the NMR resonances for the ring methyl groups of isopropylmesitylene (protons H a and H b in the following structure) are two singlets at 2.25 and 2.13 with a 2 : 1 intensity ratio,...
-
Draw a variation on the rebound hydroxylation mechanism that accounts for the formation of epoxides from some alkenes by CyP450 as shown in Fig. P17.38. C=C +47 +47 Felv Fell S S. Figure P17.38...
-
find T(625). I Given the recurrence relation T(n)=7T (n/5)+ 10n for n > 1 T (1)=1 Answer: (please write your answer here, add required space if needed)
-
Overall, property taxes constitute a small piece of the macroeconomic pie in industrialized countries, but they are an important source of revenue for local governments. To what extent do local...
-
Two charges, q=43.5 nC and qB=-25.5 nC are placed as shown in the picture. y [m] + 9 B P 9A 1 0-1- x [m] What is the magnitude of the net electric field at point P? Provide your answer in N/C, use at...

Study smarter with the SolutionInn App


