Where might the following compounds have IRabsorptions? (b) (c) .Co (a) C -H-CH2CH CH2OH
Question:
Where might the following compounds have IRabsorptions?
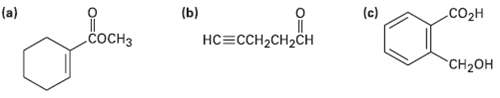
Transcribed Image Text:
(b) (c) .Coон (a) СОСНЗ нC -ссH-CH2CH CH2OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a An ester next to a double bond absorbs at 1715 cm The ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following compounds have the same molecular formulas (C4H10). Which one would you expect to have a higher boiling point?
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
At what approximate positions might the following compounds show IRabsorptions? (c) (b) CH CH (a) CH3CH2CH3 CHCCH2CH3CH2 CHCH2C3CH (f) . (d) (e) " CCH2CH2COCH CH
-
Multiple Choice 1. Which of the following concepts are pervasive in the application of generally accepted auditing standards, particularly the standards of field work and reporting? a. Internal...
-
Suppose that two firms compete in a market where consumers have identical preferences. The benefits and costs of the two firms are B 1 , C 1 and B 2 , C 2 respectively, where B 1 - C 1 > B 2 - C 2 ....
-
Calculate the formal charge on the indicated atom in each of the following molecules or ions: (a) The central oxygen atom in O3, (b) Phosphorus in PF6-, (c) Nitrogen in NO2, (d) Iodine in ICl3, (e)...
-
What is the next biggest challenge we currently face with respect to ICT and sustainability?
-
The Elon Company manufactures parts for an aircraft company using three computerized numerical controlled (CNC) turning centers. The company wants to decide which machines are capable of producing a...
-
How do transformational and transactional leadership styles influence organizational culture and employee morale, and in what contexts might each be more effective?
-
Recall that very satisfied customers give the XYZ-Box video game system a rating that is at least 42. Suppose that the manufacturer of the XYZ-Box wishes to use the 65 satisfaction ratings to provide...
-
The IR spectrum o phenyl acetylene is shown in figure. What absorption bands can youidentify? 100 80 60 40 -C=CH 20 4000 1500 Wavenumber (cm) 3500 3000 2500 2000 1000 500 Transmittance (%)
-
Where might the following compound have IR absorptions?
-
Determine whether the given point is in the feasible set of the furniture manufacturing problem. The inequalities are as follows. 1. (8, 7) 2. (14, 3) 3. (9, 10) 4. (16, 0) S6x + 3y s 96; Ix+ ys 18;...
-
What are the arithmetic and geometric average returns for a stock with a sample of annual returns of 4%, 9%, -6%, and 18%?
-
Williamson, Inc., has a debt-equity ratio of 3. The firm's weighted average cost of capital is 12 percent, and its current cost of equity is 18 percent. Williamson has no preferred stocks in its...
-
Toyo Ventures Holdings Berhad has a share price at RM26.70 and the company is expected to pay a dividend of RM0.80 per share in August 2021.The exercise price is announced at RM28.50. The risk-free...
-
Wellington Seafood Company would like to sell 4,000 shares of stock to acquire its competitor Kapiti Crab Inc. The manager decides to use Dutch auction underwriting. The bids received are: Quantity...
-
Compare and contrast the investments from a risk / reward perspective over the last five years for Amazon, Microsoft, Apple, Pfizer, and Wells Fargo. What do you notice about the average of the...
-
Out looking for Tarzan, Jane swings on a vine to speed her search. (a) During her are downward, how much work is done on her by the tensile force in the vine? (b) At which position along the arc is...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
The vapor pressure of a 1 M ionic solution is different from the vapor pressure of a 1 M nonelectrolyte solution. In both cases, the solute is nonvolatile. Which set of diagrams best represents the...
-
Predict the major products of the following reactions. (a) 1-methylcyclohexene + aqueous Hg(OAc)2 (b) The product from part (a), treated with NaBH4 (c) 4-chlorocycloheptene + Hg(OAc)2 in CH3OH (d)...
-
Show how you would accomplish the following synthetic conversions. (a) But-1-ene 2-methoxybutane (b) 1-iodo-2-methylcyclopentane 1-methylcyclopentanol (c) 3-methylpent-1-ene 3-methylpentan-2-ol...
-
(a) Count the elements of unsaturation in the three structures shown above (parsalmide, ethynyl estradiol, and dynemicin A). (b) Draw structural formulas of at least two alkynes of each molecular...
-
The Devon Motor Company produces automobiles. On April 1, the company had no beginning inventories, and it purchased 6,290 batteries at a cost of $95 per battery. It withdrew 5,800 batteries from the...
-
Potter Industries has a bond issue outstanding with an annual coupon of 6% and a 10-year maturity. The par value of the bond is $1,000. If the going annual interest rate is 8.6%, what is the value of...
-
Varsity Furniture began June with merchandise inventory of 41 sofas that cost a total of $29,110. During the month, the company purchased and sold merchandise on account as follows: June 7 June 14...

Study smarter with the SolutionInn App


