Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
Question:
Which of these compounds would you expect to be more soluble in water? Explain?
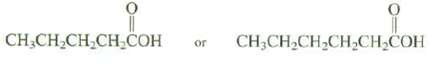
Transcribed Image Text:
CH₂CH₂CH₂CH₂COH or CH3CH₂CH₂CH₂CH₂COH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Both compounds have a polar hydrophilic part and a nonpolar hydroph...View the full answer

Answered By

Joan Gakii
I'm a meticulous professional writer with over five years writing experience. My skill set includes
- Digital Content,
- Interpersonal Communication,
- Web Content and academic Writing,
- Proofreading,
- Editing,
- Project Management, and
- Public Relations.
5.00+
7+ Reviews
12+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Which alkyl halide would you expect to be more reactive in an SN2 reaction with a given nucleophile? In each case, you can assume that both alkyl halides have the same stability a. CH3CH2CH2Br or...
-
Which alkyl halide would you expect to be more reactive in an E2 reaction? a. b. c. d. CH3 CHCHCHCH.CH, or CH.CHCH CHCH. Br Br Br CH3CH2CH CHCH3 or CH3CH2CHCH CH3 CH2CHCH2CH3 or CH2CH CHCH3
-
What types of decisions are made by football managers? Would you characterize these decisions as structured or unstructured problems? Explain.
-
Distinguish between lag and lead indicators. Provide two examples of each to illustrate your answer?
-
In the Excel file Facebook Survey, find the average and median hours online/ week and number of friends in the sample using the appropriate Excel functions. Compute the midrange and compare all...
-
Water flows under a sluice gate in a channel of 10 -ft width. If the upstream depth remains constant at \(5 \mathrm{ft}\), plot a graph of flowrate as a function of the distance between the gate and...
-
Nasreen Company issues $2 million, 10-year, 8% bonds at 97, with interest payable on July 1 and January 1. (a) Prepare the journal entry to record the sale of these bonds on January 1, 2014. (b)...
-
Applying analytical synthesis of mechanisms, design a four-bar mechanism to move the object shown in the figure, through the three positions indicated in their numbered order without taking into...
-
Sketch vo for each network of Fig. 2.176 for the input shown. 20 V Ideal v Ideal -20 V
-
Which of these isomers would you expect to have the higher boiling point: Explain? CH 3 CH 2 CH 2 OH or CH 3 CH 2 OCH 3
-
Which functional group is present in each of thesecompounds? a) CHCHCHCHOCH, ) CHCH,CH,CO,H Careful: how are the O's bonded to the C? e) CHCOCH, b) d) f) OH 0 CNH, H
-
At the end of 2013, Blackhorse Productions, Inc., used the aging of accounts receivable method to estimate that its Allowance for Doubtful Accounts should be $19,750. The account had an unadjusted...
-
There were 970 movies made in Hollywood between 2007 and 2013. Suppose that, for a documentary about Hollywood film crews, a random sample of 5 of these movies will be selected for in-depth...
-
An immersion heater operating at \(1000 \mathrm{~W}\) is in the form of a rectangular solid with dimensions of \(16 \mathrm{~cm}\) by \(10 \mathrm{~cm}\) by \(1 \mathrm{~cm}\). Determine the heat...
-
Perhaps one of the most successful public/private partnerships between a municipality and a private entity is the USTA Billie Jean King National Tennis Center. The National Tennis Center, located in...
-
Armoring Military Planes During the Second World War, the U.S. military collected data on bullet holes found in B-24 bombers that returned from flight missions. The data showed that most bullet holes...
-
As a customer, think back to a recent sales presentation that you felt went well. What made it good? What did the salesperson do (or not do) that most impressed you? Did you buy the product or...
-
Webster, Inc., dealt in automobile accessories at wholesale. Although it manufactured a few items in its own factory, among them windshield wipers, Webster purchased most of its inventory from a...
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
Find the area under the parabola y = x 2 from 0 to 1.
-
4-Chloropyridine undergoes reaction with dim ethylamine to yield 4-dimethylaminopyridine. Propose a mechanism for thereaction. CI N(CH3)2 HN(CH3)2 HCI N.
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
How does culture relate to political and security issues in the world? Please provide specific examples. 2 What are the most significant contemporary security issues in the world? How does culture...
-
Tree Seedlings has the following current-year purchases and sales for its only product. Units Acquired at Cost Date January 1 Activities Beginning inventory January 3 Sales February 14 February 15...
-
A resistor of an unknown resistance is placed in an insulated container filled with 0.50 kg of water. A voltage source is connected in series with the resistor and a current of 1.2 amps flows through...

Study smarter with the SolutionInn App


