Which of these two salts would your expect to be more soluble in hexane(C6H14)? NH, CI +
Question:
Which of these two salts would your expect to be more soluble in hexane(C6H14)?
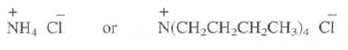
Transcribed Image Text:
NH, CI + Or N(CH₂CH₂CH₂CH3)4 Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The salt with the nonpolar hydr...View the full answer

Answered By

Palash Ghorai
Not much experience but try to solve every problem.Try my best
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which alkyl halide would you expect to be more reactive in an SN2 reaction with a given nucleophile? In each case, you can assume that both alkyl halides have the same stability a. CH3CH2CH2Br or...
-
Which alkyl halide would you expect to be more reactive in an E2 reaction? a. b. c. d. CH3 CHCHCHCH.CH, or CH.CHCH CHCH. Br Br Br CH3CH2CH CHCH3 or CH3CH2CHCH CH3 CH2CHCH2CH3 or CH2CH CHCH3
-
Which alkyl halide would you expect to be more reactive in an SNI solvolysis reaction? CH2CH3 CH3 or - CH CHCH H CH CH2CH H
-
You are asked to evaluate the following two projects for Boring Corporation using the NPV method combined with the PI approach, which project would you select? Use a discount rate of 10 percent....
-
The onstt.t0 Construction Company is a real estate developer and building contractor. The company has two sources of long-term capital: debt and equity. The cost of issuing debt is the after-tax cost...
-
Using the Excel file Facebook Survey, determine if the mean number of hours spent online per week is the same for males as it is for females.
-
Consider the flow down a prismatic channel having a rectangular cross section of width \(b\). The channel bottom makes an angle \(\theta\) with the horizontal. Show that \[ \frac{d y}{d x}=\frac{\tan...
-
The following expenditures relating to plant assets were made by Devereaux Company during the first 2 months of 2014. 1. Paid $5,000 of accrued taxes at time plant site was acquired. 2. Paid $400...
-
The United World Corporation has three subsidiaries. Sales Net income (after taxes) Assets Computers $20,690,000 2,160,000 21,220,000 Magazines $5,770,000 668,000 8,780,000 Cable TV $4,590,000...
-
A plane wave traveling along the x-axis in a polystyrene-filled region with e, = 2.54 has an electric field given by E, = Ee cos(ot - kx). The frequency is 2.4 GHz, and Eo = 5.0 V/m. Find the...
-
Explain which compound you expect to have the higher boiling point. a) cont b) CH3CHCOH c) CH,CH,OCH,CH, d) CH H or or CHCHCHCH or CHCH,CH,OCH,CH,CH, or H
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
Write an equation for each parabola. Each parabola is a translation of the graph of the parent function y = x2. d. 10 5 -10 10
-
In each of the following situations, indicate the statistical analysis you would conduct and the appropriate test or test statistic that should be used. a. Consumer preferences for Coca-Cola were...
-
What are the deliverables and milestones of a software development project?
-
If an investor commits $4,500 to an IRA each year for 30 years and receives 6% interest, what will her total investment be worth at the end of the 30 years?
-
Could an include use case have an include use case? Provide an example.
-
Name the strongest benefit and the greatest weakness of agile development methodologies.
-
Write a letter to Grandpa explaining why we now say that light is not just a particle, and not just a wave, but in fact is botha wavicle! 1 1 Frequency Period period' frequency
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
If f is the function whose graph is shown in Figure 2 and g(x) = 0 x f(t) dt, find the values of g(0), g(1), g(2), g(3), g(4), and g(5). Then sketch a rough graph of g. Figure 2 y -2. 1 0 1 2 y =...
-
Acid-catalyzed hydrolysis of a 1, 2-epoxycyclohexane produces a trans-diaxial 1, 2diol. What product would you expect to obtain from acidic hydrolysis of cis-3-tert-butyl-1, 2-epoxycyclohexane?...
-
Grignard reagents react with oxetane, a four-membered cyclic ether, to yield primary alcohols, but the reaction is much slower than the corresponding reaction with ethylene oxide. Suggest a reason...
-
Treatment of trans-2-chlorocyclohcxanol with NaOH yields 1, 2-epoxycyclo- hexane, but reaction of the cis isomer under the same conditions yields Cyclohexanone. Propose mechanisms for both reactions,...
-
Draw the shear diagram for the beam. Set P = 600 lb, a = 5 ft, b = 7 ft. Draw the moment diagram for the beam.
-
Ahmed, Bassem and Jamal are partners in Tadamon Company who share profits and losses at a ratio of 2 : 2 : 4 respectively. On 1 / 1 / 2 0 2 2 , it was decided to liquidate the company, and the...
-
Draw the shear diagram for the beam. Follow the sign convention. Draw the moment diagram for the beam. Follow the sign convention.

Study smarter with the SolutionInn App


