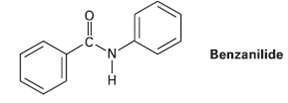
At what position, and on what ring, would you expect bromination of benzanilide to occur? Explain by
Question:
At what position, and on what ring, would you expect bromination of benzanilide to occur? Explain by drawing resonance structures of theintermediates.
Transcribed Image Text:
Benzanilide 'N' H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
00 1z Deactivated Activated by C0 byN Br2 FeBr3 H 00 Br Br ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
By drawing resonance structures for the carbocation intermediates, show why aromatic substitution in pyridine - N - oxide occurs at the 4 - position rather than at the 3 - position.
-
At what position and on what ring do you expect nitration of 4-bromo- biphenyl to occur? Explain, using resonance structures of the potentialintermediates. 4-Bromobiphenyl Br
-
Would you expect the reactivity of a five-membered ring ether such as tetrahydrofuran (Table 12.2) to be more similar to an epoxide or to a noncyclic ether?
-
What is your perspective on changes in cultural ideals for the female body from that of Marilyn Monroe in the 1950s to top models today?
-
Rex and Agnes Harrell purchased a beach house at Duck, North Carolina, in early 2015. Although they intended to use the beach house occasionally for recreational purposes, to help pay the mortgage...
-
Consider a population of 1,024 mutual funds that primarily invest in large companies. You have determined that , the mean one-year total percentage return achieved by all the funds, is 8.20 and that...
-
A surprise count of the \(Y\) Company's imprest petty cash fund, carried on the books at \(\$ 5,000\) was made on November \(10,19 X 0\). The company acts as agent for an Express Company in the...
-
Why might companies today develop IS operational plans that cover only one year?
-
This week's discussion question has three different options for your topic. YOU ONLY NEED TO DISCUSS ONE OF THEM. 1. Consider having analyzed the materials provided in State v. Mitchell as a...
-
As a fraud expert asked to investigate possible fraud at a local nonprofit organization, you suspect that one of the workers, Stacey, has been embezzling money. After securing enough evidence to be...
-
At what position, and on what ring, would you expect the following sub stances to undergo electrophilicsubstitution? (a) (b) CH3 Br (c) (d) .CI CH3 Z O=U
-
Would you expect the FriedelCrafts reaction of benzene with (R)-2-chloro- butane to yield optically active or racemic product explain.
-
What is a membrane potential?
-
Explain why margin (marginal) costs are considered the "glue" that binds average variable cost and average total cost? Finally, what are some industry examples that might illustrate economies of...
-
What is the final speed of a free electron accelerated from rest through a potential difference of - 1 0 4 V ? The mass of the electron is 9 . 1 1 x 1 0 - 3 1 kg . You need to express the speed in km...
-
In a slap shot, a hockey player accelerates the puck from a velocity of 7 . 1 4 m / s to 3 6 . 3 m / s in the same direction. If this shot takes 3 . 2 7 * 1 0 ^ - 2 s , calculate the distance in m...
-
Freight trains can produce only relatively small accelerations and decelerations. If the train is moving at 7 0 . 9 mph and slows at a rate of 0 . 4 3 m / s ^ 2 , how far in kilometers will it take...
-
How many feet of 3 0 - gauge wire is required to have a resistance of 1 ohm?
-
Karen Jones purchased 200 shares of Mex Inc. common stock for \($10\) per share exactly 2 years ago, in December 2014. Today, December 15, 2016, the stock is selling for \($18\) per share. Because...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
Redo Problem 5.22 using Aspen Plus. Problem 5.22 Isobutane is to be liquefied to make liquid petroleum gas (LPG). The butane is available at 25 C and 1 bar, it will be compressed to 15 bar, cooled...
-
Show syntheses of these compounds from1-bromobutane: b) TH. c)
-
Show the products of thesereactions: CH CH;CH C3D CH CH3 .. -- NaOH CH3
-
Explain how a similar hydroboration reaction could be used to prepare (R)-2-butanol in good enantiomeric excess.
-
Novak Corporation's statement of financial position as at December 31, 2023, showed the following amounts: Cash $140; Accounts Receivable $610; Land $1,050; Accounts Payable $430; Bonds Payable $470;...
-
Submit a brief summary of this case study 'Arcelor and the global steel industry' found on pages 484-501. This a brief section often read by executives or people who want a quick read on your...
-
The following cost data pertain to the operations of Montgomery Department Stores, Incorporated, for the month of July. Corporate legal office salaries $ 6 2 , 2 0 0 Apparel Department cost of sales...

Study smarter with the SolutionInn App


