At what position, and on what ring, would you expect the following sub stances to undergo electrophilicsubstitution?
Question:
At what position, and on what ring, would you expect the following sub stances to undergo electrophilicsubstitution?
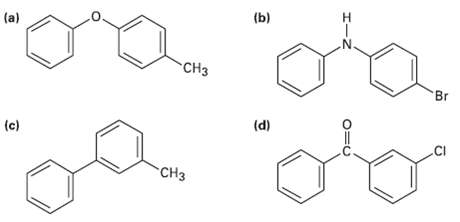
Transcribed Image Text:
(a) (b) CH3 Br (c) (d) .CI CH3 エーZ O=U
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
ogad CH3 Activated by0 Activated byO and CH3 Activated byN Substitutio...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
At what position and on what ring do you expect nitration of 4-bromo- biphenyl to occur? Explain, using resonance structures of the potentialintermediates. 4-Bromobiphenyl Br
-
Would you expect the following compound to be aromatic? Justify your answer. OR N-
-
What effect would you expect the use of MACRS depreciation rules to have on the acceptability of a project having a 10-year economic life but a 7-year MACRS classification?
-
How would this photo be different if the two people were both Americans?
-
Mona viewed herself as a creative individual who had chosen to go to law school for economic reasons. Mona's undergraduate majors were creative writing and American Indian studies. Mona was very...
-
The following substances were separated on a gel filtration column. Estimate the molecular mass of the unknown. Compound Blue Dextran 2000 Aldolase Catalase Ferritin Thyroglobulin Unknown Vr (mL)...
-
To the left of z = 0.15 and to the right of z = 0.15 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
Lucas has $ 2,000 that he wishes to invest for one year. He has narrowed his choices down to one of the following two actions: a1: Buy bonds of X Ltd., a company that has a very high debt equity...
-
Busch Gardens Sweets is preparing for Spring Break. They prepare two sizes of Peanut and Raisin Delight and need to calculate how many bags of deluxe and standard mixes they can produce. The deluxe...
-
Since LSUS corporation is producing at full capacity, Amanda has decided to have Han examine the feasibility of a new manufacturing plant. This expansion would represent a major capital outlay for...
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
At what position, and on what ring, would you expect bromination of benzanilide to occur? Explain by drawing resonance structures of theintermediates. Benzanilide 'N' H.
-
How does the volatility of a portfolio compare with the weighted average volatility of the stocks within it?
-
What is the PV of 5310 paid in year 9 if the cost of capital is 11.7(Do not round Intermediate calculations. Round your answer to 2 decimal places.) Present value. How should you calculate discus...
-
Compare the advantages and disadvantages of the existing approach with Gabriel's suggestion. Provide examples of other costs that are commonly allocated across multiple departments, plants, or...
-
For a per year additional expenditure, Rainbow can get a "Good As New" service contract that essentially keeps the machine in new condition forever. Net of the cost of the service contract, the...
-
What method of valuing professional or business goodwill looks at the net income of a professional practice or business for one year then subtracts what a reasonable salary would be for someone of...
-
In December 2011 the Consumer Prices Index stood at 121.7 (Base date: 2005). The Average Weekly Earnings index for that month for workers in the Transport and storage sector was 148.7 (base date:...
-
Which investment approach is the most likely basis for Sardars buy recommendation for AZ Industrial? A. Relative value B. High-quality value C. Deep-value investing Jack Dewey is managing partner of...
-
In a nonmagnetic medium, E = 50 cos (10 9 t 8x) a y + 40 sin (10 9 t 8x) a z V/m find the dielectric constant r and the corresponding H.
-
Redo Problem 5.23 using Aspen Plus. Problem 5.23 Repeat the calculation of problem 5.22 with the vapor being recycled to the compressor Problem 5.22 Isobutane is to be liquefied to make liquid...
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Show preparation of these alcohols fromalkenes. b) c)
-
Show the products of thesereactions: 1) BH3, THF 2) H2O2, NaOH a) Ph- b) 1) disiamylborane 2) HO2, NaOH
-
What is the minimum quantity firms need to sell in order to start earning profit given insurance costs of $50,000, materials o per unit, and break even quantity is 1500?
-
Assume a 10% discount rate and compute the present value of Rs. 1100, Rs.900, Rs.1500 andRs.700 received at the end of 1-4 years In the above sum if the amounts are received at the beginning of the...
-
Recall that an FX rate XXXYYY = Bid/Offer gives the rate of YYY per XXX. You can buy XXX/sell YYY at the offer price and sell XXX/buy YYY at the bid price. EURUSD is trading at 1.4760/1.4763 USDJPY...

Study smarter with the SolutionInn App


