Compound A, C 8 H 10 O 2 , has an intense JR absorption at 1750 cm
Question:
Compound A, C8H10O2, has an intense JR absorption at 1750 cm?1 and gives the 13C NMR spectrum shown. Propose a structure for A.
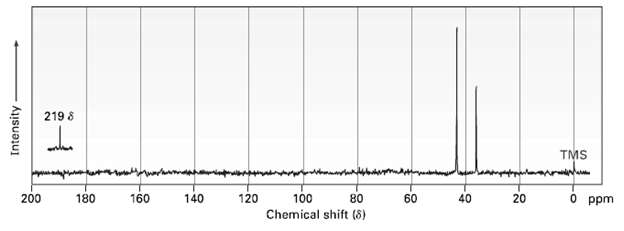
Transcribed Image Text:
219 8 TMS 200 180 160 140 120 40 O ppm 80 60 20 100 Chemical shift (8) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Compound A has 4 degrees of unsat...View the full answer

Answered By

Akash Goel
I am in the teaching field since 2008 when i was enrolled myself in chartered accountants course
Since then i have an experience of teaching of class XI, XII, BCOM, MCOM, MBA, CA CPT.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Does the 13C NMR spectrum shown in Figure 22.10 correspond to that of 1-amino-2 methyl- 2-propanol or to 2-amino-2-methyl-1-propanol? Could this compound be prepared by reaction of an epoxide with...
-
Does the 13C NMR spectrum shown in Figure 22.10 correspond to that of 1-amino-2-methyl-2-propanol or to 2-amino-2-methyl-1-propanol? Could this compound be prepared by reaction of an epoxide with...
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
SAE 10 oil at 20C flows in a vertical pipe of diameter 2.5 cm. It is found that the pressure is constant throughout the fluid. What is the oil flow rate in m3/h? Is the flow up or down?
-
Why is SAS rated as a top company to work for by Forbes? How does SAS attract and keep top talent?
-
Jessica put in an order for some shares of Exxon Mobil Corp. a. As shown on the ticker, how many shares did Jessica buy? b. How much did each share cost? c. What was the value of Jessica's trade? HD...
-
Graphically depict, label, and describe the operations within the System Capability Construct (Figure 10.17). Failed Initial State Deactivated Activate Activated Activated Pre-Capability Operations...
-
Jason Elsner started his own consulting firm, Elsner Company, on June 1, 2014. The trial balance at June 30 is shown below. In addition to those accounts listed on the trial balance, the chart of...
-
Oakmont Company has an opportunity to manufacture and sell a new product for a four-year period. The company's discount rate is 18%. After careful study, Oakmont estimated the following costs and...
-
Since LSUS corporation is producing at full capacity, Amanda has decided to have Han examine the feasibility of a new manufacturing plant. This expansion would represent a major capital outlay for...
-
Propose structures for molecules that meet the following descriptions. Assume that the kinds of carbons (1, 2, 3, or 4) have been assigned by DEPT-NMR. (a) C 6 H 12 O; IR: 1715 cm 1 ; 13 C NMR: 8.0 ...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 4 H 7 C1O ??IR: 1715 cm ?1 ? (b) C 7 H 14 O ? ? ?IR: 1710 cm ?1 ? (c) C 9 H 10 O 2 ? ? ?IR: 1695 cm ?1 ?...
-
Explain yield to maturity in terms of the spot rate.
-
Miguel is an accounting manager for Bronx Shoes Company. In designing the coding scheme for the companys accounting system, Miguel is considering assigning numbers A10 through A99 for all asset...
-
What are the ethical considerations surrounding resilience promotion, particularly in vulnerable populations, and how can interventions be designed to empower individuals without inadvertently...
-
The height of a person above the ground on a Ferris wheel is given by the function: 1 H (t) = 9 sin - 20 +12 2 where H(t) is the height in metres of the person t seconds after taking a seat on the...
-
The lost value of equipment over a period of time is called depreciation. The simplest method for calculating depreciation is straight-line depreciation. The annual straight-line depreciation D of an...
-
Orders have been coming in like crazy since the special order bundle sale started! Aaliyah has asked Missy to help them out with packing and shipping everything. Together, they were able to get the...
-
The Jacobi identity is a requirement of the commutation relations of a Lie algebra that ensures the corresponding Lie group is associative. For elements \(\hat{A}, \hat{B}, \hat{C}\) of a Lie...
-
What are the 5 Cs of marketing channel structure?
-
The molecule H 2 O 2 has a resultant dipole moment of 2.2 D. Can this molecule be linear? If not, describe a shape that might account for this dipole moment.
-
Show how you might prepare each of the following amines through reductive amination: (a) (b) (c) NH2 CH3 CH3
-
When phenyl isothiocyanate, C6H5N == C == S, is reduced with lithium aluminum hydride, the product formed has these spectral data: MS (m/z): 107, 106 IR (cm-1): 3330 (sharp), 3050, 2815, 760, 700 1H...
-
When N,N'-diphenylurea (A) is reacted with tosyl chloride in pyridine, it yields product B. The spectral data for B include: MS (m/z): 194 (M+.) IR (cm-1): 3060, 2130, 1590, 1490, 760, 700 1H NMR...
-
Describe one product development recommendation and one market development recommendation for Tesla. Explain why this idea should be adopted by the Tesla EV company in the New Zealand market.
-
A U.S. investor is considering investing $100 million in Fiat stock in Italy. The stock has an expected return of 5% in euros, including the dividend yield, and the investor is forecasting a 3%...
-
Determine "Tyson Food "Company's firmographics, activities, and objectives. Then relate these differences to differences in the organizational cultures of the organizations. or provide detail of any...

Study smarter with the SolutionInn App


