Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 4
Question:
Propose structures for ketones or aldehydes that have the following 1H NMR spectra:
(a) C4H7C1O ??IR: 1715 cm?1?
(b) C7H14O ? ? ?IR: 1710 cm?1?
(c) C9H10O2? ? ?IR: 1695 cm?1?
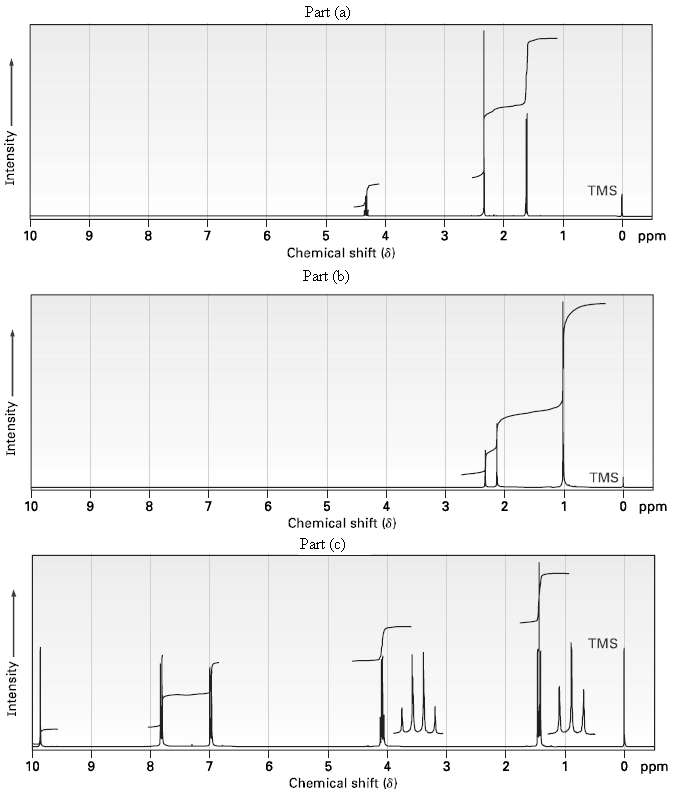
Transcribed Image Text:
Part (a) TMS 0 ppm 10 4 3 Chemical shift (8) Part (b) TMS O ppm 10 6. Chemical shift (8) Part (c) TMS O ppm 10 7. Chemical shift (8) Intensity Intensity Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
OC As always calculate the degree of unsaturation f...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
Is the PS assessment a valid predictor of performance as a store manager? Would you recommend the PS be used in the future to select sales people for promotion to store manager?
-
Assume you were the manager of a social enterprise like Greenling. How would you go about attracting individuals to work for your organization?
-
Phil sold his shares of Verizon Communications Inc, as indicated on the above ticker. a. How many shares did he sell? b. How much did each share sell for? c. What was the total value of all the...
-
What is the System Capability Construct?
-
On October 1, 2012, Faith Schultz established Heavenly Realty, which completed the following transactions during the month: a. Faith Schultz transferred cash from a personal bank account to an...
-
Q6: Capacity Planning - Capacity Levels (3 Marks) If you know that Zayed's branch operations at PRIMO's Pizza are designed to operate 16 hours a day. In addition, across the whole year, there will be...
-
from a locally grown plant. By 2020, the initiatives were shown to be not commercially viable even though support was received from the Biotech Oil Engineering Association and Environmental society....
-
Compound A, C 8 H 10 O 2 , has an intense JR absorption at 1750 cm ?1 and gives the 13 C NMR spectrum shown. Propose a structure for A. 219 8 TMS 200 180 160 140 120 40 O ppm 80 60 20 100 Chemical...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 10 H 12 O ? ??IR: 1710 cm ?1 ? (b) C 6 H 12 O 3 ? ? ?IR: 1715 cm ?1 ? (c) C 4 H 6 O ? ? ? ?IR: 1690 cm ?1 ?...
-
Graph each system of equations and find any solutions. Check your answers. Identify the system as consistent or inconsistent. If the system is consistent, state whether the equations are dependent or...
-
Tegan is a commission sales person. In October, Tegan sold $50,000 in product. What is the value of Tegan's commission? The commission rates are: 2% for the first $10,000 4% for the next $15,000 ...
-
A clothing company sells ski jackets every winter but must decide in the summer how many jackets to produce. Each jacket costs $65 to produce and ship and sells for $129 at retail stores. For the...
-
A ceiling fan turns at a rate of 20 RPM. A 1 gram bug hangs on to the end of a 50 cm fan blade. What is the magnitude of the frictional force between the bug's feet and the blade?
-
How might emerging technologies such as virtual reality therapy and biofeedback training be utilized to facilitate resilience-building by providing immersive, personalized experiences that promote...
-
Betty Corporation issued $ 4 4 0 , 0 0 0 of 4 % , 1 0 - year bonds on January 1 , 2 0 2 4 , for $ 4 0 5 , 7 0 4 . This price provided a yield of 5 % on the bonds. Interest is payable semiannually une...
-
The Killing form of a Lie algebra provides the definition of normalization of operators in a particular representation of the Lie algebra. For representation \(R\) of \(\mathfrak{s u}(2)\), the...
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
Which of the following molecules would you expect to be polar? (a) HCN; (b) SO 3 ; (c) CS 2 ; (d) OCS; (e) SOCl 2 ; (f) SiF 4 ; (g) POF 3 . Give reasons for your conclusions.
-
Propose a mechanism that can explain the occurrence of this reaction: 0 CH2
-
When acetone is treated with anhydrous ammonia in the presence of anhydrous calcium chloride (a common drying agent), crystalline product C is obtained on concentration of the organic liquid phase of...
-
The difference in positive-charge distribution in an amide that accepts a proton on its oxygen or its nitrogen atom can be visualized with electrostatic potential maps. Consider the electrostatic...
-
Wimble Ltd had $400 million of debt outstanding at an interest rate of 9% and $600 million of equity (market value) outstanding. Wimble is subject to a 30% corporate tax rate. What is the amount of...
-
explain and comments thies pints Has existed for a long time One of the original fast-food establishments High standing Commonly recognized for its root beer. With high-quality cuisine Markets to...
-
12 Stiler XYZ currently has an enterprise value of $600 million, 20 million shares outstanding, $200 million in excess cash and no debt. Assuming XYZ uses its excess cash to repurchase shares, and...

Study smarter with the SolutionInn App


