Predict the major product of each of the following reactions: (b) (a) -C CH HCI Ether HCI
Question:
Predict the major product of each of the following reactions:
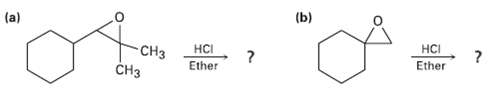
Transcribed Image Text:
(b) (a) -CНз CHз HCI Ether HCI Ether
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
Strategy As discussed in this section acidcatalyzed epoxide ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
Predict the major product of each reaction below: (a) (b) (c) 1) EtMgBr 2) H20 :? H. 1) PhMgBr 2) H20
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
The Apache NetBeans IDE is mainly composed of NetBeans Open-Source IDE and NetBeans Platform. (True/False)
-
What are some of the greatest IT challenges for the coming year?
-
The operations manager for an appliance manufacturer wants to determine the optimal length of time for the washing cycle of a household clothes washer. An experiment is designed to measure the effect...
-
Enumerate the steps in using (a) ratio and (b) difference estimation.
-
1. How did Hastings change his use of communications in sending and receiving messages from Pure Software to Netflix? 2. How did Hastings change his use of feedback from Pure Software to Netflix? 3....
-
a company knows that it will need to pay $750,000 each year for the next 10 years to clean up toxic pollution at a farmer plant site. Management wants to set aside a lump sum now that will be enough...
-
Using the information given below, prepare an income statement and an owner's equity statement for the year 2019 for Rapid Car Services from the adjusted trial balance. Owner Stella Grafton did not...
-
Reaction of cis-2-butene with m-chloroperoxybenzoic acid yields an epoxide different from obtained by reaction of the trans isomer. Explain.
-
How would you prepare the following diols? (b) (a)
-
Consider the following argument: In a framework that allows uncertain initial stares, disjunctive effects are just a notational convenience, not a source of additional representational power. For any...
-
Applying a Theorist ( J ane Addams, 'Utilization of Women in City Government') to a current piece of Pop Culture Using the ideas from a specific theorist, first concisely elaborate the core ideas...
-
Discussion Question1 Go to the US Department of Homeland Security webpage: "Take the Challenge", located at: https://www.dhs.gov/see-something-say-something/take-challenge On this webpage, there are...
-
Why do you think alcohol is the mostly widely abused drug in the United States? How can we better communicate the dangers and challenges associated with heavy and/or sustained alcohol use? According...
-
If a firm's production process is Q = 0.8L2 -0.2L, and labor costs $95 per worker and output sells for $5, how much labor should the firm hire?
-
In light of the multifaceted challenges posed by the rise of hunger, unemployment, and poverty in America, as discussed in Alan Shapiro's "The Rise of Hunger, Unemployment Unemployment & Poverty in...
-
As of December 31, 2025, Stoneland AG has assets of 3,500 and equity of 2,000. What are the liabilities for Stoneland AG as of December 31, 2025? a. 1,500. b. 1,000. c. 2,500. d. 2,000.
-
Make an argument that Williams had a right to delay the closing until after August 1.
-
A stream of hot water at 85 C and a rate of 1 kg/s is needed for the pasteurizing unit in a milk-bottling plant. Such a stream is not readily available, and will be produced in a well-insulated...
-
Determine the structure of this compound from its IR and 13C-NMR spectra, its formula isC7H16O2: C,H1,0, 80- 60- %T 40 20- 0- 500 1000 1500 2000 2500 3500 3000 4000 Wavenumber (cm) C,H1602 All CH's...
-
Predict the multiplicities of the indicated hydrogen's in the 1H-NMR spectra of these compounds: CH,CI I H I - + CH,CI CI - c) b) --- I H I - ()
-
Predict the approximate chemical shifts, multiplicities and integrals for the absorptions in the 1H-NMR spectra of thesecompounds: CH2 b) .- COCH CH3 a) d) CH;CHCH CH3 c) CH;CH,NHCH,CH3
-
Image transcription text Wind tunnel measurements of the pressure and skin friction around a NACA 2415 airfoil at 8 degrees angle of attack resulted in the following data of pressure and skin...
-
Image transcription text The following table contains load-extension data from a tensile test on a cylindrical specimen with gauge length 9mm and gauge diameter 5mm. Load-extension Data Load [KN] 0...
-
Image transcription text Systems Modelling and Analysis - Assignment 1 Due: Friday 25/08/2022 by 5:00:00 pm. To be submitted individually on Canvas and Gradescope. Part 1: Dartboard Positioning...

Study smarter with the SolutionInn App


