Provide names for thesecompounds: b) a) , CI Br - d) ) e) CH3
Question:
Provide names for thesecompounds:
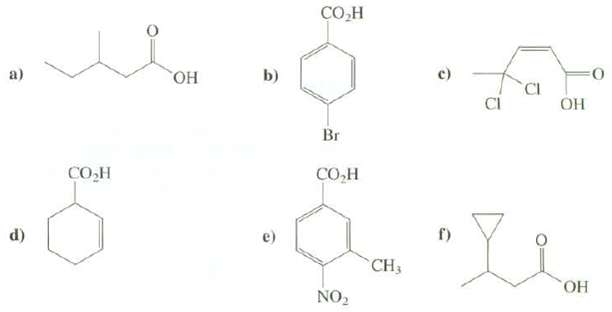
Transcribed Image Text:
СОн b) a) но, CI ОН Br СОН СО-Н d) г) e) CH3 ОН NO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a 3Methaylpentanoic acid b pBrom...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Provide IUPAC names for the following compounds. (a) (CH3)2CHCH2CH3 (b) CH3-C(CH3)2-CH3 (c) (d) (e) (f) CH CH CHCH CH,CHCHCH le ' CH CH,CH, CH CH CH, CH CH C(CH CH,CH,CHCHCH, CH(CH2 CH CHCH,CH, CH)C...
-
Provide at least five different acceptable IUPAC names for the following compound.
-
Provide the full names for the following acronyms or abbreviations used in thischapter. OCR CPU MFLOPS UPC DASD DVD DBMS ICL HTML OOP 4 GL CASE
-
8. Describe each of the following types of isomerism (i) geometrical isomerism (2 marks) (ii) solavate isomerism (2 marks) (iii) coordination isomerism (2 amrks)
-
One division of a large defense contractor manufactures telecommunication equipment for the military. This division reports that 12% of non-electrical components are reworked. Management wants to...
-
A beam of protons with various speeds is directed in the positive x direction. The beam enters a region with a uniform magnetic field of magnitude 0.52 T pointing in the negative z direction, as...
-
The current price of gold is \(\$ 412\) per ounce. The storage cost is \(\$ 2\) per ounce per year, payable quarterly in advance. Assuming a constant interest rate of \(9 \%\) compounded quarterly,...
-
Shop Rite Services is ready to prepare its financial statements for the year ended December 31, 2012. The following information can be determined by analyzing the accounts: 1. On August 1, 2012, Shop...
-
a. If my car needs $1,200 in repairs today, do I have enough to cover the cost? b. If my job frequently requires me to work late or on weekends, is it worth it to pay additional money for premium...
-
Consider a particle is confined in a three-level system. The probability of finding the particle in first level is 0.38, for second level 0.36, and for third level it is 0.26. What is the entropy of...
-
(a) Is sodium hydroxide a strong enough base to completely remove a proton from the ?-carbon acetone; that is, does this equilibrium lie nearly completely to the right when sodium hydroxide is the...
-
Draw structures for these compounds: (a) 6-Bromo-3, 5-dichlorohexanoic acid (b) Cyclobutanecarboxylie acid (c) m-Chlorobenzoic acid (d) (E)-3-Phenyl-2-propeonic acid
-
If you wanted to store the US stock markets total capitalization (roughly $31 trillion at the close of fiscal year 2022) as whole dollars, what primitive data type could you use?
-
Which of the following answers best describes a downburst? Group of answer choices A local severe down draft of air that results in strong winds aloft Funnels of wind whose speeds reach over 300 mph...
-
Charges of -5.10 C and -3.60 4C are placed 3.00 cm apart as shown: 3.00 cm -5.104C Charge A 7 3.48 108 N/C 6.00 107 N/C 5.22' 106 N/C 8.99' 105 N/C P Charge B Refer to the above diagram. The...
-
A composite plane wall consists of a Li= 50 mm thick layer of insulation (ki= 0.05 W/m K) and a Ls= 25 mm thick layer of siding (ks- 0.10 W/m-K). The inner temperature of the insulation is 20C. The...
-
The average horse eats 7.515 kilograms of hay per day. An average bale of hay costs around $5.50 and weighs around 262,050 decigrams. If a farm has 100 horses, what is the cost per year to feed them?...
-
You have won the lottery: The BernLotto offers you the following options for getting your payment: Option 1) Get a payment of 2 million at the end of years 1 and 2 and then 1.5 million every year...
-
In late 2012, an article in the Wall Street Journal, observed that: The once-predictable Chinese yuan has become increasingly volatile, a development that . . . poses greater foreign-exchange risks...
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
From the point of view of preventing or reducing global warming, does it matter where on the planet the reduction in global-warming gases takes place?
-
Fats can be either optically active or optically inactive, depending on their structure. Draw the structure of an optically active fat that yields 2 equivalents of stearic acid and 1 equivalent of...
-
Spermaceti, a fragrant substance from sperm whales, was much used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester...
-
The plasmalogens are a group of lipids found in nerve and muscle cells. How do plasmalogens differ fromfats? CH2OCH=CHR CR A plasmalogen H2OR"
-
Suppose a, b, and c are int variables and a = 5 and b = 6. What value is assigned to each variable after each statement executes? If a variable is undefined at a particular statement, report UND...
-
In terms of hydrogen ion concentration, how much more acidic is a solution with a pH of 4.0 than a solution with a pH of 5.5? Round your answer to the nearest tenth. Show Recall that pH, p(x), is...
-
The following was submitted under the original assignment of "write small program that uses dynamic binding".. now what is requested is to suggest another way to take advantage of the dynamic biding...

Study smarter with the SolutionInn App


