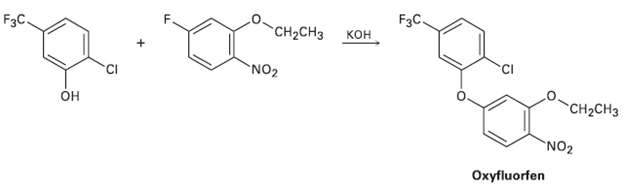
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism.
Question:
The herbicide oxyfluorfen can be prepared by reaction between a phenol and an aryl fluoride. Propose amechanism.
Transcribed Image Text:
FзC. кон F3C. "CH-CHз NO2 он "CH-CHз NO2 Охyfluorfen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (19 reviews)
Hydroxide is used to form the nucleophilic phenoxide anion F3C F3C F...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following alkyl bromide can be prepared by reaction of the alcohol (S)-2-pentanol with PBr3. Name the compound, assign (R) or (S) stereochemistry, and tell whether the reaction of the alcohol...
-
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism. NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H DMSO HO F3C F3C CI FC...
-
A compound known as Hagemann's ester can be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid and heat. Write the structure for the product of...
-
Calculate e y for the following values of y: (a) y = 1 (b) y = 5 (c) y = -.5 (d) y = -2.5 (e) y = 3.1 (f) y = - I (g) y = .05 (h) y = .32 (i) y = 6.1 (j) y = -5.4
-
Lonzo owns two apartment buildings. He acquired Forsythia Acres on February 21, 1998, for $300,000 ($90,000 allocated to the land) and Square One on November 12, 2016, for $800,000 ($100,000...
-
Consider a reflection grating operating with an incident angle of 40 in Figure 19-7. (a) How many lines per centimeter should be etched in the grating if the first-order diffraction angle for 600-nm...
-
Pregnancy Length Use the normal distribution in Exercise 15. (a) What percent of the new mothers had a pregnancy length of less than 280 days? (b) What percent of the new mothers had a pregnancy...
-
Review your results from S22-3. Grippers expects cost of goods sold to average 60% of sales revenue, and the company expects to sell 4,100 pairs of shoes in March for $260 each. Grippers target...
-
What role does process simulation play in the optimization of energy and material efficiency in chemical processes, and how can simulation tools help identify bottlenecks and opportunities for...
-
The debits and credits from four related transactions, (1) through (4), are presented in the following T accounts. Assume that the freight terms were FOB shipping point and that the credit terms were...
-
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4 (b) (a)
-
Treatment of p-bromotoluene with NaOH at 300 oC yields mixture of two products but treatment of m-bromotoluene with NaOH yields a mixture of three products. Explain.
-
Use polar coordinates to find the limit. Let x = r cos and y = r sin , and note that (x,y) (0, 0) implies r 0. lim (x, y)(0, 0) cos(x + y)
-
How should the timeframe of end of fiscal year October 31, 2022 be explained in the presentation?
-
1. You have access to $1,500,000 or 1,000,000. Assume the spot rate is $1.50/. 2. The annualized 4-month interest rate is 6.5% in US and 5.1% in the UK. 4-month forward rate is quoting 1.48 US dollar...
-
What makes perpetuities unique, and how does the infinite nature of perpetuities impact their value over time?
-
Alpha company is trying to install a new machine that has estimated cost Rs. 200,000 by replacing an old machine. The new machine required installation cost Rs. 20,000. The expected increase in...
-
What type of team is made up of people pulled from various departments, but they still have to do their departmental job in addition to being on the innovation team?
-
Which of Tongs statements regarding equity index benchmarks is (are) correct? A. Only Statement 1 B. Only Statement 2 C. Both Statement 1 and Statement 2 Evan Winthrop, a senior officer of a US-based...
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
By measuring the temperature change accompanying a differential volume change in a free expansion across a valve and separately in a reversible adiabatic expansion, the two derivatives (T/ V ) H and...
-
Explain which mechanism is preferred in these reactions and show the majorproducts: Br b) 1-BUOH 0- PROH Br . EIOH, reflux OTs d) DMF + CH,CO + KOH Ph Br Br . ELOH ELOH f) e) + CH,CH,0
-
Explain which reaction mechanism (E1, SN1, E2, SN2) these reactions follow, and show the majorproducts: Br a) DMSO + CN Br b) . reflux + OH Br CH c) EIO LOTS + NaOEt d) t-BUOH CI + t-BUOK e) . Br CH,...
-
Show all the steps in the mechanism for this reaction. What substitution product(s) would also be formed in thisreaction? Br E:OH
-
Image transcription text Wind tunnel measurements of the pressure and skin friction around a NACA 2415 airfoil at 8 degrees angle of attack resulted in the following data of pressure and skin...
-
Image transcription text The following table contains load-extension data from a tensile test on a cylindrical specimen with gauge length 9mm and gauge diameter 5mm. Load-extension Data Load [KN] 0...
-
Image transcription text Systems Modelling and Analysis - Assignment 1 Due: Friday 25/08/2022 by 5:00:00 pm. To be submitted individually on Canvas and Gradescope. Part 1: Dartboard Positioning...

Study smarter with the SolutionInn App


