Using the data below, calculate the partial molar enthalpies of 1-propanol and water as a function of
Question:
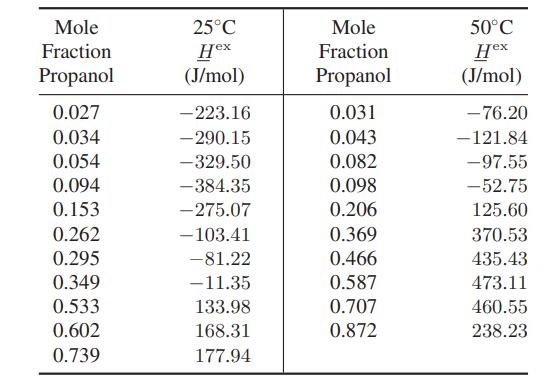
Using the data below, calculate the partial molar enthalpies of 1-propanol and water as a function of composition at both 25°C and 50°C.
data: V. P. Belousov, Vent. Leningrad Univ. Fiz., Khim, 16(1), 144 (1961).
Transcribed Image Text:
Mole Fraction Propanol 0.027 0.034 0.054 0.094 0.153 0.262 0.295 0.349 0.533 0.602 0.739 25°C Hex (J/mol) -223.16 - 290.15 -329.50 -384.35 -275.07 -103.41 -81.22 -11.35 133.98 168.31 177.94 Mole Fraction Propanol 0.031 0.043 0.082 0.098 0.206 0.369 0.466 0.587 0.707 0.872 50°C Hex (J/mol) - 76.20 -121.84 -97.55 -52.75 125.60 370.53 435.43 473.11 460.55 238.23
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To calculate the partial molar enthalpies H at a given temperature and composition you can use the f...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
A partial molar property of a component in a mixture may be either greater than or less than the corresponding pure-component molar property. Furthermore, the partial molar property may vary with...
-
Use the data in problem 8.29 to compute the partial molar enthalpies of pyridine and acetic acid in their mixtures at 25C over the whole composition range. Problem 8.29 Two streams containing...
-
Jessica took out a $19,000 demand loan at 7% simple interest.She paid off the loan with a $2600 payment in 4 months, a $8000 payment in 10 months and a final payment in 15 months. Find the size of...
-
10 1 Journalize the following transactions for Bigelow Company for the month of October. 31 Oct Stockholders invest cash in the company in exchange for common stock. 5 Oct The company buys a delivery...
-
David files a bankruptcy petition under Chapter 13. After the claims of secured and priority creditors have been satisfied, Davids remaining bankruptcy estate has a value of $100,000. Davids...
-
Linearising the Euler equation in the consumption CAPM model results in a a linear regression model between the log returns of a risky asset and the growth rate in consumption \[...
-
Suggest a method of using the Bode plot of phase angle versus frequency (Fig. 3.11(b)) to identify the natural frequency and the damping ratio of the system.
-
The Poster Bed Company believes that its industry can best be classified as monopolistically competitive. An analysis of the demand for its canopy bed has resulted in the following estimated demand...
-
lim x-3x-10 x-2 x+4x+4 Find the following limit algebraically, if it exists.
-
XYZ is an unlevered firm and is currently valued at $820,000. It has 15,000 shares outstanding. As part of a Management Buyout (MBO), XYX is planning to borrow $400,000 from a bank at an annual...
-
An equimolar mixture of nitrogen and acetylene enters a steady-flow reactor at 25C and 1 bar of pressure. The only reaction occurring is The product leaves the reactor at 600C and contains 24.2...
-
Following are the slightly smoothed heat-of-mixing data of R. P. Rastogi, J. Nath, and J. Misra [J. Chem. Thermodyn., 3, 307 (1971)] for the system trichloromethane (component 1) and 1,2,4-trimethyl...
-
Is density a unique measure of mass distribution in a volume? Does it vary? If so, on what kind of scale (distance)?
-
During 2 0 2 4 , its first year of operations, Baginski Steel Corporation reported a net operating loss of $ 3 6 0 , 0 0 0 for financial reporting and tax purposes. During 2 0 2 5 , Baginski reported...
-
LCD Industries purchased a supply of electronic components from Entel Corporation on November 1 , 2 0 2 4 . In payment for the $ 2 5 . 0 million purchase, LCD issued a 1 - year installment note to...
-
The DeVille Company reported pretax accounting income on its income statement as follows: 2 0 2 4 $ 4 3 0 , 0 0 0 2 0 2 5 3 5 0 , 0 0 0 2 0 2 6 4 2 0 , 0 0 0 2 0 2 7 4 6 0 , 0 0 0 Included in the...
-
Suppose that the distance a car travels varies directly with the amount of gasoline it uses. A certain car uses 14 gallons of gasoline to travel 448 miles. How many miles can the car travel if it has...
-
Factor the polynomial by grouping (if possible). 5n2p-10np 5n2 +10n
-
Identify the four characteristics or features of the perils insured against by property- casualty insurance. Rank the features in terms of actuarial predictability and total loss potential.
-
Identify Thank You mission, strategy and core competencies. Identify strategy changes that have taken place at Thank You since its founding in 2008. Your answer must in text references and must be...
-
Express your height in the units of inches, feet, and meters.
-
Make sure you know how to start and quit a MATLAB session. Use MATLAB to make the following calculations, using the values x = 10, y = 3. Check the results by using a calculator. a. u = x + y b. v =...
-
Suppose that x = 2 and y = 5. Use MATLAB to compute the following. a. yx 3 /x - y b. 3x/2y c. 3/2xy d. x 5 /x 5 - 1
-
Key comparative figures for Apple and Google follow. $ millions Net sales Cost of sales 161,782 163,756 Apple Google Current Year Prior Year Current Year Prior Year $ 260,174 $ 265,595 $ 161,857 $...
-
First, use both the text and Brady v US to explain plea bargaining. What features must a guilty plea have in order to be valid? Which constitutional rights are implicated by pleading guilty? (Make...
-
Stephen is a well-known member of parliament in the (fictional) country of Costat, a contracting party to the European Convention of Human Rights. He has risen to prominence in recent years through...

Study smarter with the SolutionInn App


