The balanced equation for the reaction of gaseous nitrogen dioxide and fluorine is The experimentally determined rate
Question:
The balanced equation for the reaction of gaseous nitrogen dioxide and
fluorine is
![]()
The experimentally determined rate law is
![]()
A suggested mechanism for this reaction is
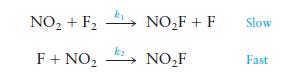
Is this an acceptable mechanism? That is, does it satisfy the two requirements?
Transcribed Image Text:
2NO(g) + F(g) 2NOF(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The first requirement for an acceptable mechanism is that the sum of ...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
3. Calculate and Explain, how much the action described has added to GDP: A company sells 50 computers at a retail price of $1000 apiece and 100 software packages at a retail price of $50 apiece to...
-
The reaction of nitric oxide (NO(g)) with molecular hydrogen (H2(g)) results in the production of molecular nitrogen and water as follows: 2NO(g) + 2H 2 (g) N 2 O(g) + 2H 2 O(g) The experimentally...
-
The dissolved oxygen present in any highly pressurized, hightemperature steam boiler can be extremely corrosive to its metal parts. Hydrazine, which is completely miscible with water, can be added to...
-
Here is the end-of-year account balance information from the accounting records of Jaunty Coffee Company: Sales revenue Cash Cost of goods sold Accounts payable Capital stock Dividends Retained...
-
(a) Use a computer algebra system to draw a direction field for the differential equation. Get a printout and use it to sketch some solution curves without solving the differential equation. (b)...
-
Use a calculator to approximate sin /8 Round the answer to two decimal places.
-
Fraud deterrence is centered on the fear of getting caught and the fear of getting punished. In your opinion, which is stronger and why?
-
White Way Inc. produces and sells theater set designs and costumes. The company began operations on January 1, 2012. The following transactions relate to securities acquired by White Way Inc., which...
-
Commencing 1 April 2022, Morgan Landscaping Pty Ltd (morgan) provided Larissa (an employee) with the use of a Volkswagen Golf car. The car cost $55,000 (including GST) and was used privately every...
-
The gas-phase reaction of chlorine with chloroform is described by the equation The rate law determined from experiment has a noninteger order: A proposed mechanism for this reaction follows: Is this...
-
Butadiene reacts to form its dimer according to the equation 2C 4 H 6 (g) C 8 H 12 (g) The following data were collected for this reaction at a given temperature: a. Is this reaction first order or...
-
Approximately 14 million Americans are addicted to drugs and alcohol. The federal government estimates that these addicts cost the U. S. economy $ 300 billion in medical expenses and lost...
-
If youre not familiar with vesting, the idea is that when a firm is launched, instead of issuing stock outright to the founders, the stock is distributed over a period of time, typically three to...
-
Briefly describe each of the five stages in the organizational life cycle.
-
Kelly Andrews has developed a new line of jewelry that has created some positive buzz among friends and some business stores in her local community. When asked by a reporter, Where do you plan to...
-
Spend some time studying PurBlu Beverages, the subject of the studentinitiated business profiled at the beginning of the chapter. In what ways have PurBlu Beverages business practices and its...
-
ModCloth founders Susan Koger and Eric Koger met in high school. Susan enjoyed shopping for vintage clothing, and over time accumulated so many outfits that she decided to sell some. Eric, who was...
-
In June 1988, British & Commonwealth PLC (B&C) acquired Atlantic Computers, the worlds third largest computer-leasing company. In April 1990, B&C placed Atlantic Computers into administrative...
-
Determine the volume of the parallelepiped of Fig. 3.25 when (a) P = 4i 3j + 2k, Q = 2i 5j + k, and S = 7i + j k, (b) P = 5i j + 6k, Q = 2i + 3j + k, and S = 3i 2j + 4k. P
-
Draw Lewis structures for each of the following species and predict the hybridization at each carbon atom: (a) H 2 CCH ; (b) H 2 CCH 3 + ; (c) H 3 CCH 2 .
-
There are three isomers of difluoroethene, C 2 H 2 F 2 , which differ in the locations of the fluorine atoms. (a) Which of the forms are polar? (b) Which has the largest dipole moment? F F C= 1 C O H...
-
Explain why the lattice energy of lithium chloride (861 kJ mol -1 ) is greater than that of rubidium chloride (695 kJ mol -1 ), given that they have similar arrangements of ions in the crystal...
-
Kevin is refinishing his rusty wheelbarrow. He moves his sandpaper back and forth 4 5 times over a rusty area, each time moving a total distance of 0 . 1 5 m . Kevin pushes the sandpaper against the...
-
Metabolizing a candy bar releases about 1 0 ^ 6 joules of energy. Assume the candy bar's mass is 0 . 3 6 kg and use the formula kinetic energy = 1 / 2 mv ^ 2 , where m is the object's mass and v is...
-
Hind's Corporation has provided the following production and total cost data for two levels of monthly production volume. The company produces a single product. Production volume Direct materials....

Study smarter with the SolutionInn App


