For each molecular geometry, list the number of total electron groups, the number of bonding groups, and
Question:
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom.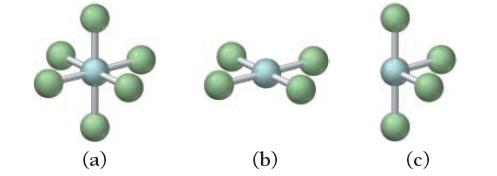
Transcribed Image Text:
နာ (a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Total electron groups 4 Bonding groups 4 Lone pairs 0 ...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom. (a) (b) (c)
-
Describe a hybridization scheme for the central S atom in the molecule SF 4 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the S atom are involved in overlaps,...
-
Describe a hybridization scheme for the central Cl atom in the molecule ClF 3 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the Cl atom are involved in...
-
finish this The five components, which are scope, references, configuration item, baseline, and verification, are vital to ensure the configuration management plan establishes control, ensures...
-
All future costs are relevant in decision making. Do you agree? Why?
-
Resolve the force F2 into components acting along the u and v axes and determine the magnitudes of the components.
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Belden, Inc. acquires 30 percent of the outstanding voting shares of Sheffield, Inc. on January 1, 2017, for $312,000, which gives Belden the ability to significantly influence Sheffield. Sheffield...
-
18) A physician prescribes one 0.7 mg tablet of a medication to be taken twice a day for 15 days. How many total mcg of the medication will the patient have taken after 15 days?
-
Determine the electron geometry, molecular geometry, and idealized bond angles for each molecule. In which cases do you expect deviations from the idealized bond angle? a. PF 3 b. SBr 2 c. CHCl 3 ...
-
A molecule with the formula AB 3 has a trigonal planar geometry. How many electron groups are on the central atom?
-
Hydrogen bonding is (a) The name given to the chemical bond in the HH or H 2 molecule. (b) A particularly strong type of dipolar interaction. (c) As strong as a chemical bond. (d) When hydrogen bonds...
-
Swedens Economic Expectations The Swedish economys growth rate in 2017 was better than expected, mainly because of an easy monetary policy. Growth prospects for 2018 look up as well, with...
-
What is the difference between an order for relief and an automatic stay ?
-
Discuss the implications of the unique characteristics of services for the marketing activities of services enterprises.
-
Outline the benefits and challenges associated with using influencer marketing. Highlight some of the main social media influencers relevant to your market, age group and interests. Assess the extent...
-
Explain the challenge that high unemployment and high inflation poses for Venezuelas monetary authorities. To work Problems 28 to 30, use the information that Venezuelas economy is expected to...
-
1. Was the CNET story sufficient justification for the HP boards actions? Why or why not? 2. HP Chairwoman Patricia Dunn defended the actions of the board by arguing that HPs higher standards of...
-
During the year land was revalued and the surplus reported as Revaluation surplus; and an asset costing 80,000, written down to 38,000, was sold for 40,000. Identify the cost of any non-current...
-
The member is to be made from a steel plate that is 0.25 in. thick. If a 1-in. hole is drilled through its center, determine the approximate width w of the plate so that it can support an axial force...
-
Determine the maximum normal stress developed in the bar when it is subjected to a tension of P = 2 kip. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
Determine the maximum axial force P that can be applied to the bar. The bar is made from steel and has an allowable stress of Ï allow = 21 ksi. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
In this numerical project, we observe Corollaries 3.16 and 3.17 in action. Consider n = 1,..., 20. For each n, consider the dyadic partition of [0,1] given by t;= j/2". Sample a single Brownian path...
-
Thomas Burberry is credited with investing gabardine fabric in the 1850s. The Burberry trademark is registered in more than ninety(90) countries around the world, and the companys signature plaid...
-
You are the Strategic Planner for ABC Health, a large nonprofit healthcare system in central PA. Congratulations! Your Board of Directors and the Executive Directive are exploring a possible merger...

Study smarter with the SolutionInn App


