For each molecular geometry, list the number of total electron groups, the number of bonding groups, and
Question:
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom.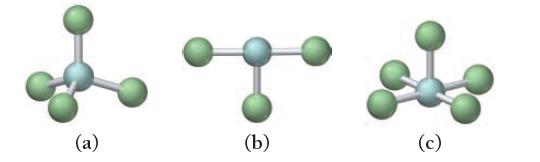
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 4 e groups b 5 e groups ...View the full answer

Answered By

Srilekha Talluri
I am a final year Computer science student at Bits Hyderabad. I have recently completed my internship at Amazon. I worked as a SDE Intern with Seller central team of Amazon. I have also done an internship with Human Resource department of Telangana Govt Secretariat. Due to these exposure i had the opportunity of putting my subject into action and make an impact.
I have also done good projects in my Discipline and worked as teaching assistant for few important Computer science courses like Data Structures and Algorithms, Discrete Mathematics. As a computer science student, I have completed lot of courses like Cryptography, Graph theory, Design and Analysis of Algorithms etc.
My teaching experience has been awesome throughout .I have had many occasions to experience teaching. I have been fortunate enough to teach underprivileged children as a part of a NGO on college. I taught them the essential aspects of writing, mathematics,science etc. They were able to excel in the board exams they undertook that year and got themselves admitted to XII class.
The experience was wonderful and it caught me by surprise that many of the students never realize their true potential and just need a little external help to overcome huge barriers in academics.
Working as a teaching Assistant for disciplinary courses also helped me to gain good knowledge and tutoring experience
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom. (a) (b) (c)
-
Describe a hybridization scheme for the central S atom in the molecule SF 4 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the S atom are involved in overlaps,...
-
Describe a hybridization scheme for the central Cl atom in the molecule ClF 3 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the Cl atom are involved in...
-
Growth Strategy A. Organic Growth This section describes how JB Hi-Fi Australia can take advantage of various organic growth strategies, including new product development, market development and...
-
Sunk costs are easy to spottheyre simply the fixed costs associated with a decision. Do you agree? Explain.
-
The 500-lb force acting on the frame is to be resolved into two components acting along the axis of the struts AB and AC. If the components of force along AC are required to be 300 lb, directed from...
-
Consider the calculation of an external rate of return (ERR). The positive cash flows in the cash flow profile are moved forward to \(t=n\) using what value of \(i\) in the \((F \mid P i, n-t)\)...
-
The Stone River Textile Mill was inspected by OSHA and found to be in violation of a number of safety regulations. The OSHA inspectors ordered the mill to alter some existing machinery to make it...
-
x-5 and justify your answer. Respond in the space 3x-1 f(x)= Determine the range of provided below.
-
Determine the electron geometry, molecular geometry, and idealized bond angles for each molecule. In which cases do you expect deviations from the idealized bond angle? a. PF 3 b. SBr 2 c. CHCl 3 ...
-
A molecule with the formula AB 3 has a trigonal planar geometry. How many electron groups are on the central atom?
-
Why is it impossible to mix candy worth $4 per lb and candy worth $5 per lb to obtain a final mixture worth $6 per lb?
-
When is a security interest perfected?
-
Why is insurable interest so important?
-
Can the Bank of Korea achieve price stability and economic growth simultaneously? The Bank of Koreas monetary policy is to reduce the vulnerability of South Korean won and achieve price stabilization...
-
What are the two ways that a corporation may undergo dissolution?
-
Bundesbanks Weidmann Opposes ECBs QE Jens Weidmann, president of Bundesbank, Germanys central bank, believes that the European Central Banks (ECB) monetary expansion policy could do more harm than...
-
Divide into groups of three or four. Each group must map out its ideal model for corporate governance of an organizationfor example, the number of people on the board of directors, separate roles of...
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
The A-36 steel plate has a thickness of 12 mm. If Ï allow = 150 MPa, determine the maximum axial load P that it can support. Calculate its elongation, neglecting the effect of the fillets. r= 30...
-
If the allowable normal stress for the bar is Ï allow = 120 MPa, determine the maximum axial force P that can be applied to the bar. 5 mm 40 mm 20 mm r = 10 mm 20 mm
-
The assembly consists of two posts AB and CD each made from material 1 having a modulus of elasticity of E 1 and a cross-sectional area A1, and a central post EF made from material 2 having a modulus...
-
On March 1 of the current year, Spicer Corporation compiled information to prepare a cash budget for March, April, and May. All of the company's sales are made on account. The following information...
-
How do emergent social paradigms, such as post-capitalism and post-colonialism, inform contemporary discourses on social change, and what implications do they have for global transformation ?
-
Potato Chips Inc. paid a dividend of $1.9 per share yesterday. You expect the dividend to grow steadily at a rate of 3% per year. What's the value of a share of the firm? Assume an interest rate of...

Study smarter with the SolutionInn App


