Write balanced complete ionic and net ionic equations for each reaction. a. KSO4(aq) b. NH4Cl(aq) c. AgNO3(aq)
Question:
Write balanced complete ionic and net ionic equations for each reaction.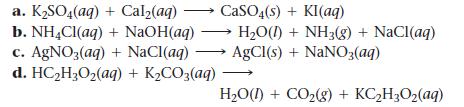
Transcribed Image Text:
a. K₂SO4(aq) b. NH4Cl(aq) c. AgNO3(aq) d. HC₂H₂O₂(aq) + K₂CO3(aq) + Cal₂(aq) + NaOH(aq) + NaCl(aq) CaSO4(s) + KI(aq) H₂O(1) + NH3(g) + NaCl(aq) AgCl(s) + NaNO3(aq) H₂O(1) + CO₂(g) + KC₂H₂O₂(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
here are the balanced complete ionic and net ionic equations for ...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HI(aq) + RbOH(aq) b. HCHO(aq) + NaOH(aq) c. HCHO(aq) + LiOH(aq)
-
Write balanced complete ionic and net ionic equations for each acidbase reaction. a. HBr(aq) + NaOH(aq) b. HF(aq) + NaOH(aq) C. HCHO(aq) + RbOH(aq)
-
Write balanced complete ionic and net ionic equations for each reaction. a. HCl(aq) + LiOH(aq) HO(1)+ LiCI(aq) b. Cas(aq) + CuCl(aq) CuS(s) + CaCl(aq) c. NaOH(aq) + HCHO(aq) d. Na3PO4(aq) + NiCl(aq)...
-
What Codification citation indicates how unrealized gains or losses from changes in fair value affect net income when investments are classified as available-for-sale?
-
The Mark Goodwin Resort is an elegant summer resort located in a remote mountain setting. Guests visiting the resort can fish, hike, go horseback riding, swim in one of three hotel pools, or simply...
-
The concept of earnings quality has several dimensions, but two characteristics often dominate: the accounting information should be a fair representation of performance for the reporting period, and...
-
Picture Gallery, Inc. works weddings and prom-type parties. The balance of retained earnings was \($18,000\) at December 31, 2010. At December 31, 2011, the businesss accounting records show these...
-
David Roberts began an upholstery cleaning business on August 1 and engaged in the following transactions during the month: Aug. 1 Began business by depositing $30,000 in a bank account in the name...
-
If you invest $20,000 at 7%, what will the value of your investment be after annual compounding? (8 marks) i) 5 years ii) 7 years iii) 12 years iv) 20 years
-
Mercury(I) ions (Hg 2 2 + ) can be removed from solution by precipitation with Cl . Suppose that a solution contains aqueous Hg 2 (NO 3 ) 2 . Write complete ionic and net ionic equations for the...
-
Write a molecular equation for the precipitation reaction that occurs (if any) when each pair of aqueous solutions is mixed. If no reaction occurs, write NO REACTION. a. Sodium chloride and lead(II)...
-
Find an equation of the tangent plane at the given point. f(x, y) = xy + xy, (-1,2)
-
ExplaintheroleofProcessControlBlock.DemonstratethechangeofinformationinPCB(Process control block) of one process from initialization to completion
-
(a) Draw a fuzzy network for F. (b) Find all routes of the fuzzy network. (d) (e) (c) Find all simple routes of the fuzzy network. Find all maximal routes of the fuzzy network. Use the fundamental...
-
Let (an)nez be a sequence of real numbers. Recall the 2(N), as a vector space over R, has the norm given by 2||(alle) (n=1 lan2)1/2 = lim N-(N=121/2 We will often denote this by llanlle to have a...
-
Fill out the rest of this table Concentration Runs 3-5 Temperature Runs 3, 6 - 8 0.2 M KI 0.2 M KNO3 0.25% starch 0.010 M Na2S2O3 0.2 M (NH4)2SO4 0.1 M EDTA Water 0.2 M (NH4)2S2O8 Time,s Temperature,...
-
On January 1, 2025, Vaughn Company purchased a new machine for $4190000. The new machine has an estimated useful life of nine years, and the salvage value was estimated to be $140000. Depreciation...
-
You must choose between the two projects whose cash flows are shown below. The projects have the same risk. a. Compute the internal rate of return and the net present value for the two projects....
-
Write a program that initializes an array. It inputs a value from the user and searches the number in the array.
-
The maximum pressure that can be developed for a certain fluid power cylinder is 5000 psi. Compute the required diameter for the piston if the cylinder must exert a force of 20 000 lb.
-
The maximum pressure that can be developed for a certain fluid power cylinder is 15.0 MPa. Compute the required diameter for the piston if the cylinder must exert a force of 30 kN.
-
A line of fluid power cylinders has a range of diameters in 1.00-in increments from 1.00 to 8.00 in. Compute the force that could be exerted by each cylinder with a fluid pressure of 500 psi. Draw a...
-
#1 What can our generation do to finally end gender discrimination within the workplace? #2 How much better would women perform in the workplace without putting half of their focus on being...
-
Goal Kick Sports (GKS) is a retail chain that sells youth and adult soccer equipment. The GKS financial planning group has developed a spreadsheet model to calculate the net discounted cash flow of...
-
On October 15, 2024, Jackson entered into a tentative agreement to sell the assets of one of its divisions. The division qualifies as a component of an entity as defined by GAAP. The division was...

Study smarter with the SolutionInn App


