Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) KCO3(aq)
Question:
Balance the following equations, and then classify each as a precipitation, acid–base, or gas-forming reaction.
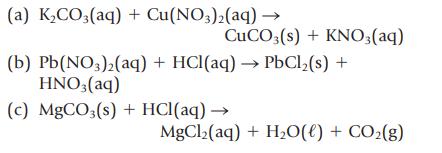
Transcribed Image Text:
(a) K₂CO3(aq) + Cu(NO3)2(aq) → CuCO3(s) + KNO3(aq) (b) Pb(NO3)2(aq) + HCl(aq) → PbCl₂(s) + HNO3(aq) (c) MgCO3(s) + HCl(aq) → MgCl2(aq) + H2O(l) + COz(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
balance and classify each of the given chemical equations a KCOaq CuNOaq CuCOs ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) Ba(OH)(aq) + HCl(aq) BaCl,(aq) + H,O(l) (b) HNO3(aq) + COCO3(s) Co(NO3)2(aq) +...
-
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acidbase, or gas-forming reaction. Show states for the products (s, , g, aq), give...
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
Gems Co. uses the indirect method to prepare its statement of cash flows. The following comparative statement of financial position for 2021 and 2022 are presented: At December 31 2022 2021 Property,...
-
How would you expect a fall in a countrys population to alter its aggregate money demand function? Would it matter if the fall in population were due to a fall in the number of households or to a...
-
A corporation donates a valuable painting from its private collection to an art museum. Which of the following are incremental cash flows associated with the donation? a. The price the firm paid for...
-
We continue the analysis begun in Exercise 1.7 by examining the relation between y = LIFEEXP and x = FERTILITY, shown in Figure 2.12. Fit a linear regression model of LIFEEXP using the explanatory...
-
1. What type of coaching function was reflected in Rowes meeting with Busche? 2. In terms of effectiveness on a 1-10 scale, with 1 being poor and 10 being excellent, what score would you assign to...
-
Cuttlefish use jet propulsion to move rapidly. To do this, a cuttlefish fills its muscular mantle cavity with water and then quickly expels the water through a tube - like opening. The mass of a...
-
Which two of the following reactions are oxidation reduction reactions? Explain your answer briefly. Classify the remaining reaction. (a) CdCl(aq) + NaS(aq) CdS(s) + 2 NaCl(aq) (b) 2 Ca(s) + O(g) 2...
-
In the following reactions, decide which reactant is oxidized and which is reduced. Designate the oxidizing agent and the reducing agent. 2+ (a) CrO (aq) + 3 Sn+ (aq) + 14 H3O+ (aq) 2 Cr+ (aq) + 3...
-
a. Show that the cubic polynomials P(x) = 3 2(x + 1) + 0(x + 1)(x) + (x + 1)(x)(x 1) And Q(x) = 1 + 4(x + 2) 3(x + 2)(x + 1) + (x + 2)(x + 1)(x) Both interpolate the data b. Why does part (a) not...
-
Assume a Hotelling line of a distance l = 1. There are two companies A and B, both of them being located at a longer distance. Company A is placed closer to the end of the line, while company B is...
-
4. Asian women in the US are more likely to study in college and more likely to study a science major than Latino women. a. Science majors have many more male students than female students. If most...
-
This is an Industrial Organization Economics Question: Suppose that demand for rollerblades is given by D(p) = A ? p. The cost function for all firms is C(y) = wy2 + f , where f is a fixed set-up...
-
Suppose you are stranded on an island. Luckily, this island is rich in clams and mangos. If you devote all of your time to harvesting clams, you can get 100 clams in a week. If you us all of your...
-
Year Quarter Units 1 Q1 20 1 Q2 100 1 Q3 175 1 Q4 13 2 Q1 37 2 Q2 136 2 Q3 245 2 Q4 26 3 Q1 75 3 Q2 155 3 Q3 326 3 Q4 48 4 Q1 92 4 Q2 202 4 Q3 384 4 Q4 82 5 Q1 176 5 Q2 282 5 Q3 445 5 Q4 181 Using...
-
Does a statistics course improve a students mathematics skills, as measured by a national test? Suppose a random sample of 13 students takes the same national mathematics examination just prior to...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
Predict the products that are expected when each of the following compounds is heated with concentrated HBr. a. b. c. d.
-
Draw all constitutionally isomeric ethers with molecular formula C 4 H 10 O. Provide a common name and a systematic name for each isomer.
-
discusses concepts of social costs and benefits, and private costs and benefits in the context of business activities from these sources: References: 1. Tietenberg, T., & Lewis, L. (2016)....
-
The following financial information is taken from the balance sheets of the Lambeth Company and the Maritza Company: Lambeth Maritza Total debt $350,000 $850,000 Total assets 550,000 1,000,000...
-
(Calculating changes in net operating working capital) Duncan Motors is introducing a new product and has an expected change in net operating income of $290,000 Duncan Motors has a 33 percent...

Study smarter with the SolutionInn App


