Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) Ba(OH)(aq)
Question:
Balance the following equations, and then classify each as a precipitation, acid–base, or gas-forming reaction.
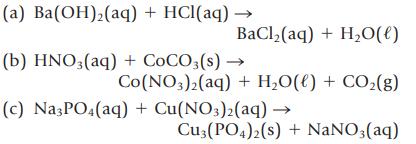
Transcribed Image Text:
(a) Ba(OH)₂(aq) + HCl(aq) → BaCl,(aq) + H,O(l) (b) HNO3(aq) + COCO3(s) → Co(NO3)2(aq) + H₂O(l) + CO₂(g) (c) Na3PO4(aq) + Cu(NO3)2(aq) → Cu3(PO4)2(s) + NaNO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
To balance and classify each of the given chemical reactions well first balance the equations and ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Balance the following equations, and then classify each as a precipitation, acidbase, or gas-forming reaction. (a) KCO3(aq) + Cu(NO3)2(aq) CuCO3(s) + KNO3(aq) (b) Pb(NO3)2(aq) + HCl(aq) PbCl(s) +...
-
Balance equations for these reactions that occur in aqueous solution, and then classify each as a precipitation, acidbase, or gas-forming reaction. Show states for the products (s, , g, aq), give...
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
Suppose that the following equations describe an economy: C = 170 + 0.60YD M s = 735; P = 1 T = 200 I = 100 - 4i Md = 0.75Y - 6i G = 350 a) what is the equation for equilibrium in the goods market?...
-
Suppose there is a reduction in aggregate real money demand, that is, a negative shift in the aggregate real money demand function. Trace the short-run and long-run effects on the exchange rate,...
-
In the article "When News Reporters Deceive: The Production of Stereotypes" (Journalism & Mass Communication Quarterly, Vol. 84, No. 2, pp. 281-298), researchers D. Lasorsa and J. Dai investigated...
-
List applications of I.C. engines.
-
Jessica James is considering a business ventureselling custom embroidered baseball caps from a pushcart kiosk at College Mall. The caps will be available in 12 different colors and one-size fits all....
-
What are three resource management tools that facilitate family decision making? Discuss how to structure a successful performance appraisal interview?
-
Which two of the following reactions are oxidation reduction reactions? Explain your answer briefly. Classify the remaining reaction. (a) CdCl(aq) + NaS(aq) CdS(s) + 2 NaCl(aq) (b) 2 Ca(s) + O(g) 2...
-
In the following reactions, decide which reactant is oxidized and which is reduced. Designate the oxidizing agent and the reducing agent. 2+ (a) CrO (aq) + 3 Sn+ (aq) + 14 H3O+ (aq) 2 Cr+ (aq) + 3...
-
The S&P 500 Index is currently at 3,000. You manage a $15 million indexed equity portfolio. The S&P 500 futures contract has a multiplier of $50. a. If you are temporarily bearish on the stock...
-
Solve the inequality:-n
-
1. Compute cost of goods manufactured.
-
The entire graph of the function h is shown in the figure below. Write the domain and range of h using interval notation. 3 m 3 4
-
Simplify 98 to the form ab.
-
GM is trying to reinvent the Cadillac brand with new car designs and a new brand story. They commission an integrated marketing communications campaign using several media. A key component of their...
-
Company S is an 80%-owned subsidiary of Company P. Company S needed to borrow $500,000 on January 1, 2011. The best interest rate it could secure was 10% annual. Company P has a better credit rating...
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
Starting with cyclohexene and using any other reagents of your choice, show how you would prepare each of the following compounds. a. b. c. OH OMe
-
When 1, 4-dioxane is heated in the presence of HI, compound A is obtained: a. Draw the structure of compound A. b. If one mole of dioxane is used, how many moles of compound A are formed? c. Show a...
-
Tetrahydrofuran (THF) can be formed by treating 1, 4-butanediol with sulfuric acid. Propose a mechanism for this transformation. H,SO, H;SO. 1,4-Butanediol Tetrahydrofuran (THF)
-
Key comparative figures for Apple and Google follow. $ millions Net sales Cost of sales 161,782 163,756 Apple Google Current Year Prior Year Current Year Prior Year $ 260,174 $ 265,595 $ 161,857 $...
-
First, use both the text and Brady v US to explain plea bargaining. What features must a guilty plea have in order to be valid? Which constitutional rights are implicated by pleading guilty? (Make...
-
Stephen is a well-known member of parliament in the (fictional) country of Costat, a contracting party to the European Convention of Human Rights. He has risen to prominence in recent years through...

Study smarter with the SolutionInn App


